Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
The Indian developers of an antivenom formulation designed to be taken orally, which has shown promising results in laboratory tests, are hoping to develop it into a tablet that could be taken straight after snakebites.
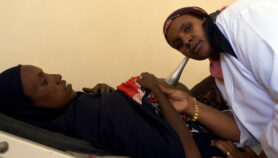
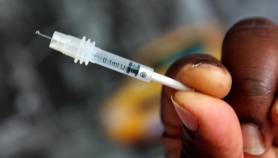
Antivenom is currently injected into patients. And since injections are often stored at hospitals that are many miles away from the rural locations where most snakebites occur, patients must usually wait for hours after being bitten before they can receive treatment. Three-quarters of patients who die from snakebites do so before they reach hospital.
“A globally initiated ‘antivenom as first aid’ can change the mortality rate markedly.”
Roshnara Mishra, University of Calcutta
Because of this, “we have felt that taking care of transit time damage will be most crucial”, says researcher Roshnara Mishra of the University of Calcutta, India, who led the team that published details of the formulation last month (7 August) in PLOS Neglected Tropical Diseases.
In the formulation, antivenom is coated in alginate, a cheap and non-toxic gum-like substance made from sugar. The researchers showed that this protected the antivenom from being digested in intestines taken from mice, and that it remained effective at neutralising snake venoms after passing through this tissue.
“A globally initiated ‘antivenom as first aid’ can change the mortality rate markedly,” says Mishra.
Matthew Lewin, a researcher at the California Academy of Sciences, United States, says: “It’s a cool idea to cloak the antivenom in alginate. Who wouldn’t want to be able to swallow something to counteract deadly venom?”
Lewin has developed a cheap antiparalytic nasal spray to treat snakebite victims.
Yet he says antivenoms can be dangerous. As they have high complication rates and, if mishandled, the side effects can be worse than the bites themselves, Lewin says they should always be given in a hospital.
Antivenom is also expensive, costing around US$70 per dose. But Mishra says that any oral formulation based on alginate, which is inexpensive, would cost little more than this.
She says her team now plans to develop the oral formulation further and eventually test it in clinical trials.
Link to full paper in PLOS Neglected Tropical Diseases