27/05/21
Health body, not trade lawyers, ‘should decide patents access’
By: Laura Owings
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
Trade negotiations are dictating access to COVID-19 vaccines, treatments and diagnostics. But will patent waivers increase access to lifesaving medicines in the global South? Laura Owings reports.
Eight months ago, South Africa and India proposed a trade-related waiver for COVID-19 vaccines, treatments and diagnostics. Calling for a temporary suspension of intellectual property protections on COVID-19 products and technologies, it was a bold move to address global inequality of access.
“As new diagnostics, therapeutics and vaccines for COVID-19 are developed, there are significant concerns [about] how these will be made available promptly, in sufficient quantities and at affordable price to meet global demand,” said the proposal, submitted to the World Trade Organization Council for Trade-Related Aspects of Intellectual Property Rights (TRIPS).
Developing countries have been disproportionately affected by the pandemic, and intellectual property rights are hindering the provision of affordable medical products to patients, the communication said.
While the proposal has gained worldwide support from Nobel laureates, international advocacy organisations and civil society, more than half of all WTO members are yet to show support. As of 17 May, 62 countries had signed on as co-sponsors, including the 35 members of the WTO’s Least Developed Countries Group.
That opposition took a meaningful shift in early May, however, when United States Trade Representative Katherine Tai announced her government’s support for waiving intellectual property protections for COVID-19 vaccines.
“The Administration believes strongly in intellectual property protections, but in service of ending this pandemic, supports the waiver of those protections for COVID-19 vaccines,” Tai said in a statement. “We will actively participate in text-based negotiations at the WTO needed to make that happen.”
“We’re facing a global public health crisis… but here we are negotiating with trade delegations, lawyers and diplomats. We need a health body making these decisions.”
Fatima Hassan, the Health Justice Initiative
The announcement was a breakthrough, drawing new supporters for the waiver including New Zealand, the European Union and France. More than 400 charities, academics and healthcare workers have petitioned the British government to also back the US proposal.
But global health equality advocates remain cautious of the US position.
“Having the US at the table means many more countries will take it seriously and join negotiations,” says Leena Menghaney, global intellectual property advisor to the Doctors Without Borders Access Campaign.
“But we must be careful about the US position. Their statement is very narrow, mentioning only vaccines when we need treatments and diagnostics as well.”
Even if the waiver were adopted immediately, however, global production of these much-needed medical supplies would still be a way off, as developing countries require substantial and critical infrastructure, capacity and skills.
Untapped capacity
With just two per cent of the world’s 700 million doses having gone to African countries and with case numbers rising in some parts of the continent, there is a particular sense of urgency surrounding vaccines.
The India-South Africa waiver proposal would allow technology transfers, knowledge sharing and logistical know-how, and enable African partners to contribute to the global COVID-19 fight.
Africa is full of untapped and underutilised capacity, says Fatima Hassan, founder of the Health Justice Initiative, based in South Africa.
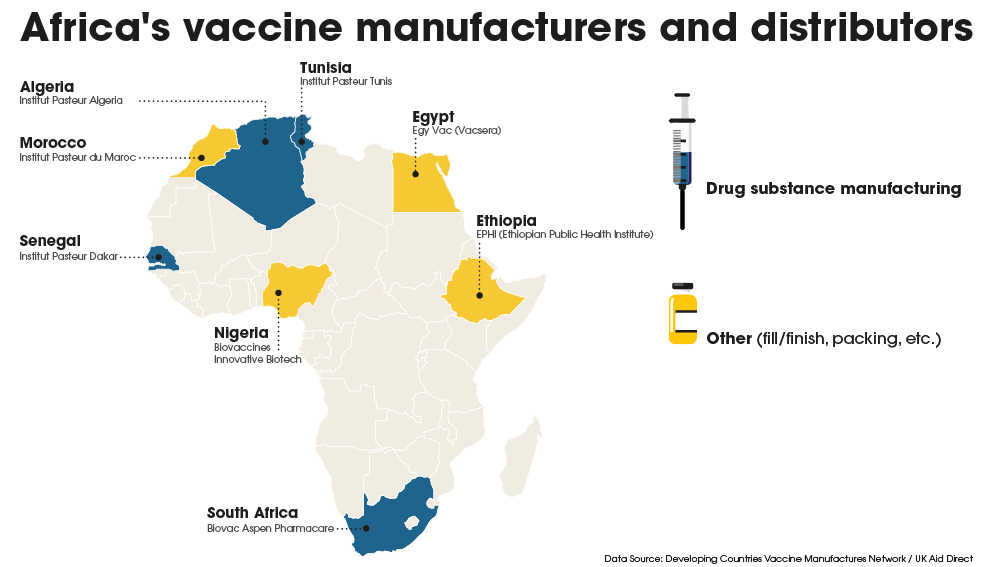
The Africa Centres for Disease Control and Prevention has identified six vaccine manufacturing sites on the continent with the potential to scale up, including two in both Nigeria and South Africa.
“Opponents say that nobody in the global South can make safe and effective vaccines. If there’s cooperation and transfer of knowledge, it’s possible,” Hassan tells SciDev.Net.
In April, the World Health Organization issued a call for expressions of interest in its mRNA technology hub, which aims to expand production capacity in low-and middle-income countries. The WHO says it has received about 50 applications from entities that are either willing to be considered as a technology transfer hub, or able to provide knowledge, training, and intellectual property rights.
Once knowledge is shared, companies can assess if they are able to retrofit or build facilities capable of manufacturing medicines and vaccines. This potential to scale up incentivises funding and investment by government.
But some say this would not have an immediate impact on COVID-19 management strategies.
“We have infrastructure that would be essential in vaccine manufacture, but would need substantial development for mRNA and live viral vector vaccines as these are new technologies,” says Salim Abdool Karim, director of the Centre for the AIDS Programme of Research in South Africa (CAPRISA).
“In South Africa, if we started now, it would take more than a year to be in a position to manufacture vaccines locally.”
More broadly across Africa, there are concerns about the ability to produce quality vaccines, says Kelly Chibale, director of the Drug Discovery and Development Centre (H3D) at the University of Cape Town. Specifically, he notes the absence of critical technical skills and technology, as well as limited local and regional regulatory capacity to ensure quality.
“However, the waiver is a step in the right direction with medium to long-term benefits in terms of acquiring and developing manufacturing,” Chibale says.
Quality control
The political will needed to ramp up vaccine production in Africa is deepening, according to analysis from consulting firm McKinsey, citing this year’s launch of the African Continental Free Trade Area (AfCFTA) and the nascent African Medicines Agency.


While there are challenges surrounding regulation of COVID-19 vaccines, Andy Gray, a pharmacist and senior lecturer at the University of KwaZulu-Natal in South Africa, says they are similar to global pressure to bring products to market quickly.
“It’s quite unusual, for example, to see trial phases one, two and three run in almost parallel, with conditional regulation given for vaccines even before phase three is complete,” he tells SciDev.Net. “Regulatory approval in general is having to be made with incomplete data.”
Arguments about the safety of vaccines should also be considered in the global context, says Gray, who is also a consultant pharmacist for the WHO Collaborating Centre on Pharmaceutical Policy and Evidence Based Practice.
Johnson and Johnson, for example, lost a potential 15 million doses of vaccine after a US subcontractor failed to meet quality standards.
In Brazil, authorities announced they were rejecting the import and use of Russia’s Sputnik V vaccine over concerns about lack of reliable data and potential health risks. Sputnik V says the move was politically motivated and contradicted an earlier decision by Brazil’s science ministry, which recognised the vaccine as safe.
“Substandard products coming out of legitimate factories but without quality control are a challenge with any new entrant into the market,” Gray says. “We should avoid labelling this as an African problem, when it can happen in the best of places.”


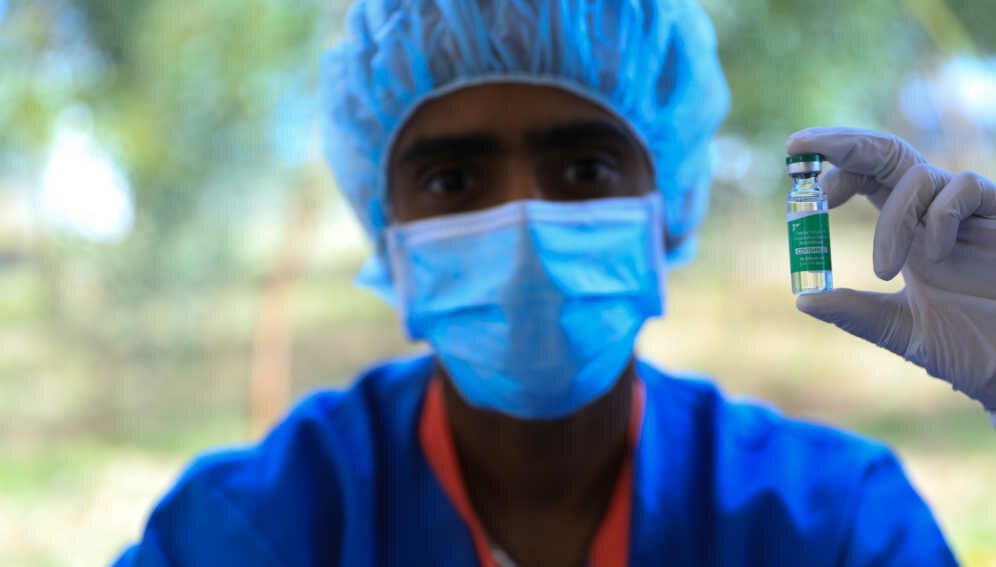
BioNTech’s mRNA COVID-19 vaccine.
Extreme emergency
With the world’s fastest growing population, and ongoing crises with HIV, measles and Ebola, Africa’s ability to produce its own vaccines could have broad public health and economic benefits. According to McKinsey, the market for African vaccines could grow from US$1.3 billion today, to between $2.3 billion and $5.4 billion by 2030.
While the TRIPS waiver could help kick-start that development, it is not intended to be a panacea for the continent.
“The intent of the waiver is to open up access to all COVID-19 related technology, including vaccines, for the duration of the pandemic,” says Hassan from the Health Justice Initiative. “It’s for a limited time period and within the rules of TRIPS.”
According to Hassan, this is not the first time a waiver has been requested and granted for a specific timeframe. And the Doha Declaration on the TRIPS agreement and public health, adopted by WTO members in 2001, specifically allows for flexibility to the TRIPS Agreement in urgent scenarios.
“[P]ublic health crises, including those relating to HIV/AIDS, tuberculosis, malaria and other epidemics, can represent a national emergency or other circumstances of extreme urgency,” the declaration says.
More than 167 million cases of COVID-19 have been confirmed worldwide, including almost 3.5 million deaths, according to the WHO. If that does not constitute extreme urgency, some epidemiologists ask, what would?
“We’re facing a global public health crisis, and all the reasons why you’d be allowed to have a waiver or use the Doha Declaration are there,” says Hassan. “It’s a real crisis moment, but here we are negotiating with trade delegations, lawyers and diplomats.”
Ultimately, Hassan says, the status quo of addressing access to lifesaving medicines and vaccines through a trade body has to change. “There’s a certain mismatch that’s not sustainable. We need a health body making these decisions.”
Focus on the future
TRIPS waiver sponsors are expected to head into text-based negotiations with WTO colleagues in June. Both Hassan and Doctors Without Borders’ Menghaney worry those will be built around a US-backed, watered-down version of the proposal.
“Our hope is that India and South Africa will hold to their original terms. That will be a big turning point, but we expect push back,” says Menghaney.
The negotiations are likely to be drawn out. As the clock ticks, some — including the Independent Panel for Pandemic Preparedness and Response, which released a damning report that said the deadly pandemic could have been avoided — have suggested setting concrete deadlines if results do not materialise.
The panel called for major vaccine-producing countries and manufacturers to convene within three months to immediately waive intellectual property rights and agree to voluntary licensing and technology transfer, if they cannot agree to do so voluntarily.
While there is no legal compulsion to this guidance, panel member Precious Matsoso tells SciDev.Net: “There is a monumental moral and ethical compulsion to save lives not in one country or a few countries, but across the globe.”
That urgency extends beyond controlling COVID-19, to preparing the world for the next pandemic threat.
“We want to see a shift to where innovation is an inclusive, truly global model aimed at delivering global public goods,” Matsoso says. “When, not if, the next pandemic arrives, we should be ready, and this global system should be functional.”