Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
[SÃO PAULO] Researchers have produced a protein that kills parasitic intestinal worms, by genetically engineering a bacterium similar to those used in probiotics — raising hopes of more effective and safer therapies for infections that affect up to two billion people worldwide.
The protein, Cry5B, has previously been shown to kill parasitic worms. It is normally produced by Bacillus thuringiensis, a bacterium used as an insecticide and not considered safe for use in people.
Bacteria containing Cry5B could be an ideal drug against human parasites, researchers say, as they can be easily and cheaply produced in large quantities, as well as shipped and stored under adverse conditions.
The researchers inserted the protein-producing gene into another related bacterium, Bacillus subtilis — strains of which are commonly used in foods such as probiotic yoghurts.
They first showed that the modified strain successfully produces the protein, and then tested it for treating parasitic worms in hamsters.
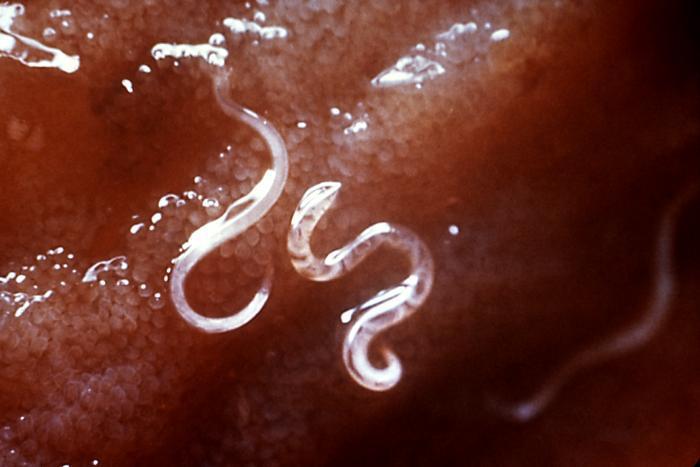
When given in small doses to hamsters infected with hookworm, Ancylostoma ceylanicum — which is capable of infecting people, and is related to a major human parasite, A. duodenale — the protein reduced the parasite burden by 93 per cent.
The study reports that this is comparable or even more effective than currently approved drugs for treating hookworms, whipworms and large roundworms.
These parasitic worms "are the leading causes of disease burden and disability in children and pregnant women worldwide" and "infect mostly impoverished people in the developing world and contribute significantly to keeping these people trapped in poverty", the study says.
Rose Gomes Monnerat, a researcher at the Brazilian Agricultural Research Corporation (Embrapa), and a member of the study team, tells SciDev.Net: "Treatment of gut parasites has been done with highly toxic drugs so far."
"There is also a growing number of reports of drug resistant parasites. So it is important to have alternatives to their control," she adds.
Manoel Victor Franco Lemos, a biologist at São Paulo State University, Brazil, says: "Although the results have been achieved by using animal models of parasitic infections, the worm species used are quite close to those that cause the same infections in humans".
But he highlights the need for trials on humans.
Raffi Aroian, co-author and a biologist at the University of California, San Diego, says: "We are talking to knowledgeable people about how much pre-clinical testing would have to be done prior to human clinical trials".
One of the main challenges, Aroian adds, is that although the B. subtilis strain used is a model for food-safe bacteria and used in some probiotics, it is not a proven food-safe bacterium.
"Now we need to put the gene into a proven food-safe one," he says.
"Additionally, several toxicity tests must also be done until we can ensure its safety," says Monnerat.
The study will be published in the September issue of Applied and Environmental Microbiology.
Link to full article in Applied and Environmental Microbiology
References
Applied and Environmental Microbiology doi:10.1128/AEM.01854-13 (2013)