Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
[CAPE TOWN] In the wake of publication of the tsetse fly’s genome, scientists say insights from the discovery are likely to open up new avenues of research into possible ways of controlling the flies and the parasitic disease they spread: sleeping sickness.
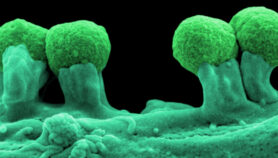
It took an international team — more than half of whom are from Africa — more than ten years to decode the genetic blueprint of Glossina morsitans, one species of tsetse fly. But the results were finally published in Science last month (25 April).
The WHO says cases of sleeping sickness, which occurs in 36 Sub-Saharan African countries, fell for the first time in 50 years to below 10,000 in 2009. The WHO aims to eradicate the disease by 2020.
Geoffrey M. Attardo of Yale University, United States, led the project. He tells SciDev.Net that charting the fly’s genome will help scientists understand its basic biology, and thus potentially design ways to control it.
“Whether such methods will significantly improve on currently available methods — in terms of cost and efficacy — is a matter of debate.”
John Hargrove, Stellenbosch University
Identifying the species’ protein-coding genes offers an exciting opportunity to understand the molecular basis for its behavioural, ecological and physiological traits, says Chris Weldon, a fly expert at the University of Pretoria, South Africa.
For example, the researchers discovered a set of visual and odour proteins that seem to drive key behavioural responses such as searching for hosts or mates.
One of these explains the tsetse fly’s attraction to the blue-black coloured cloths — a trait already widely exploited in the development of traps to reduce the disease’s spread.
The work could also open up other control strategies, says Jan Van Den Abbeele of the Institute of Tropical Medicine in Antwerp, Belgium, who led the international team’s work on the fly’s salivary gland genes. This was published in PLOS Neglected Tropical Diseases as one of several papers recently released by the project alongside the main Science one.
These methods could include, he says, producing genetically engineering and releasing males that are sterile, to keep fly numbers down. This approach was approved in Brazil last month as a means of controlling mosquitoes that transmit dengue fever.
But the discovery could open up completely new control strategies too.
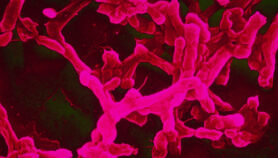
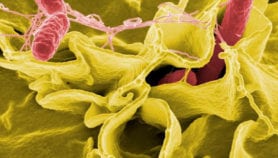
“The tsetse fly saliva is also the sole microenvironment in which the human sleeping sickness parasite develops into the infective forms that are transmitted through tsetse fly bites,” says Abbeele. He says understanding the salivary glands and the processes that support the parasite’s development in them could help to develop transmission-blocking methods.
Unusually among insects, female tsetse flies produce a type of milk to nourish their young. Since scientists have now identified the genetic instructions that help build the milk-producing cellular machinery, it may be possible to find ways to interfere with it, the researchers say.
The genomes of other tsetse fly species that transmit sleeping sickness, including G. pallidipes and G. palpalis, will be included in the next group of tsetse species to be sequenced, says Otto Koekemoer, a researcher at the Agricultural Research Council, South Africa. This will enable important aspects to be compared between the species, he says.
John Hargrove, who is director of the South African Centre for Epidemiological Modelling and Analysis at Stellenbosch University, says the possibility of new control methods was interesting. But “whether such methods will significantly improve on currently available methods — in terms of cost and efficacy — is a matter of debate,” he says.
> Link to full paper in Science
> Link to full paper in PLOS Neglected Tropical Diseases
References
Science doi: 10.1126/science.1249656 (2014)
PLOS Neglected Tropical Diseases doi: 10.1371/journal.pntd.0002649 (2014)