18/07/19
Ebola outbreak in DRC is international emergency, says WHO

Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
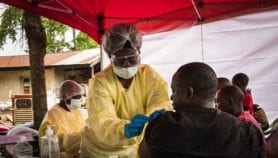
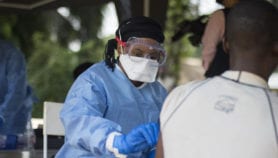
[KINSHASA] The Ebola outbreak in the Democratic Republic of Congo (DRC) is now a “public health emergency of international concern” (PHEIC), the World Health Organization (WHO) has declared, calling on the international community to step up action and funds to stem the crisis.
The announcement follows the discovery of a case in Goma, a city of almost 2 million inhabitants, where an infected pastor stayed for some time before he died. A woman has also died after crossing the border from DRC into Uganda last week to buy fish.
The declaration came late Wednesday on the recommendation of the International Health Regulations Emergency Committee, citing the development in Goma, which borders with Rwanda and has an international airport.
“Unless the international community steps up and funds the response now, we will be paying for this outbreak for a long time to come,”
Tedros Adhanom Ghebreyesus, WHO director-general
The committee stressed, however, that the decision should not mean the closure of borders and travel routes, which it said would negatively impact the emergency response and local communities.
The committee had declined on three previous occasions to invoke a PHEIC – the WHO’s highest level of alarm – saying that it did not meet all the criteria.
Since the start of the outbreak in August 2018 there have been 17, 2019 reported cases and 1,698 deaths, according to figures from the Congolese Ministry of Health.
WHO director-general Tedros Adhanom Ghebreyesus, making the announcement in Geneva, said: “It is time for the world to take notice and redouble our efforts. We need to work together in solidarity with the DRC to end this outbreak and build a better health system.”
Tedros stressed the need for unrestricted movement, saying that closing borders would only encourage people to use informal crossings that are not monitored, thereby increasing the risk of spreading the disease.
He also noted that 75 million Ebola virus screenings have already been conducted at borders and other checkpoints.
“Closing borders could have disastrous consequences for the lives and livelihoods of the people who cross the border every day for trade, education or to visit relatives,” he added.
Crisis costs
He said the PHEIC is not a measure intended to organise an international fundraiser, but about “preventing the international spread of disease”, adding that WHO is working to finalise a comprehensive strategic response plan and evaluate financial implications.
But he warned: “Unless the international community steps up and funds the response now, we will be paying for this outbreak for a long time to come.”
The strategic response plan is expected to provide the resources needed for the next phase of the response, which “will run to the hundreds of millions" [of dollars], according to the WHO chief.
The international health regulations emergency committee also expressed disappointment at what Tedros called "funding delays that limited the response ".
Declaring a PHEIC is an exceptional measure, which has only been taken four times by WHO: in 2009, during the H1N1 flu epidemic, in 2014 for polio and the Ebola epidemic which killed more than 11,300 people in West Africa, and in 2016 for the Zika virus.
The decision was broadly welcomed by the scientific community. Josie Golding, epidemics lead at UK-based charitable foundation Wellcome, said it was the “right response”.
“A step-up in the response, led by the DRC and with full international support, is critical if we are to bring the epidemic to an end. This must include enhanced diplomatic, public health, security and logistic efforts as well as releasing much needed financial resources,” she said.
Countries must not wait for the Ebola virus to spread across borders or appear on their doorsteps before taking action, she added.
Vaccine urgency
At the WHO press conference Wednesday in Geneva, Michael Ryan, executive director of the organisation's health emergency programs, stressed that accelerating vaccine delivery is a priority in the WHO’s response, amid mounting pressure to extend the coverage.
Since last year, authorities in DRC have been vaccinating people with the experimental vaccine rVSV-ZEBOV, made by international drugs company Merck.
"We support the introduction of a second vaccine, as recommended by the SAGE [Strategic Advisory Group of Experts], subject to appropriate approvals at the national level," said Ryan.
The SAGE recommended an increase in the number of people eligible for the rVSV-ZEBOV innoculation and the introduction of new experimental vaccines, including MVA-BN, developed by Johnson & Johnson and the Chinese vaccine candidate Ad5-EBOV, developed by CanSino and the Beijing Institute of Biotechnology in China.Congolese Minister of Health Oly Ilunga Kalenga, in a circular seen by SciDev.Net, said it was open to receiving proposals from experimental vaccine producers who would like to evaluate their vaccines in the DRC.
However, he added: "Given the risks of confusion, the communication problems and the confusion between the currently used rVSV-ZEBOV-GP vaccine and other vaccines that do not have the same level of evidence, the use of any other vaccine against Ebola virus disease is prohibited in the provinces of North Kivu and Ituri.”