By: Julien Chongwang
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
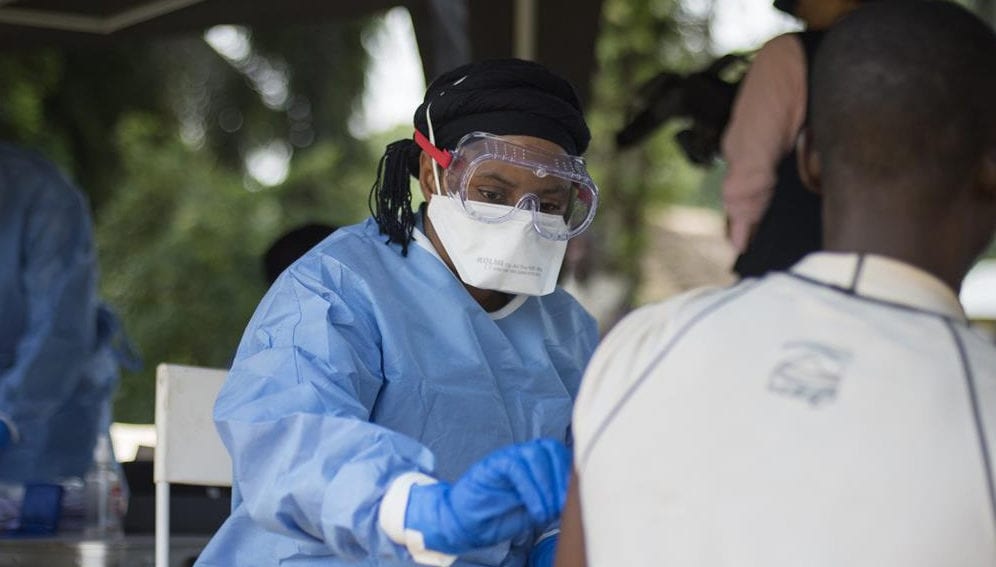
The scientific community has warned against complacency over Ebola in the Democratic Republic of Congo (DRC) after two drugs were confirmed to be effective against the virus.
The US National Institutes of Health announced last week that two of four drugs in a clinical trial running in North Kivu province were found to significantly increase survival rates.
Since the start of the outbreak in August last year, 2,877 cases of Ebola have been recorded in the DRC, of which at least 1,934 people are believed to have died.
“It will give much hope to people with Ebola who previously had no effective treatment available to them,”
Anthony Fauci, director, National Institute of Allergy and Infectious Diseases, US
Yap Boum II, a public health researcher at the University of Yaoundé I, in Cameroon, said: "Having drugs makes sense only if you can end the epidemic. It will be necessary to steer communities to refer all patients in treatment centres, so that they receive the drugs that have just been approved.”
This is a view shared by Anthony Fauci, director of the National Institute of Allergy and Infectious Diseases (NIAID), in the United States, who cautions that measures to protect populations against the disease must remain in place.
"Fighting the epidemic also requires preventing the spread of infection and this relies on good public health measures such as identification of infected people, isolation, contact tracing and administration of vaccines to contacts, and contacts of contacts, of infected people," said Fauci.
The US researcher believes that the rVSV-ZEBOV vaccine, produced by the US pharmaceutical company Merck & Co, has done much to contain the epidemic in the DRC.
However, Boum warns that a single vaccine against a disease like Ebola is not enough. He supports the ongoing testing in Uganda and soon in DRC, of the experimental vaccine MVA-BN-Filo, proposed by the US firm Johnson & Johnson.
He explained that there would be "two complementary vaccines, in that rVSV-ZEBOV is used to stop an epidemic, while MVA-BN would be used as part of a more or less long-term prevention strategy".
Aside from the availability of effective drugs, researchers stress that Ebola control must also include community engagement, surveillance, and conflict resolution to enable health care workers to reach patients and vice versa.
The ongoing conflict in the country has greatly hampered attempts to stem the outbreak, which was declared by the WHO last month to be an international emergency.
On 12 August, US National Institutes of Health announced that the current phase of trials into four drugs in the DRC would end, with all future patients to receive the two drugs found to be effective.
The two drugs in question are REGN-EB3 and mAb114, products made of monoclonal antibodies developed respectively by US laboratories Regeneron and Ridgeback Biotherapeutics.
Monoclonal antibodies
"These drugs are monoclonal antibodies,” explained Boum. “That is, they are molecules that specifically identify the virus when it is present in the body. And once these antibodies surround the virus, it is neutralised and eliminated."
Fauci told SciDev.Net: "These monoclonal antibodies are directed against an important component of the virus. They bind to the virus and neutralise it so that the infection cannot continue in the infected person.”
According to Boum, who participated in the study as a researcher at Epicenter – a program of epidemiological activities and research set up by Doctors Without Borders (MSF) – this action is also very fast.
"An infusion of mAb114, for example, will immediately allow you to see the positive evolution of the patient. And this evolution is even more immediate if the patient arrives early in the hospital. That brings its probability of healing to more than 90 per cent," he said.
Both drugs emerged from a series of trials and tests in the DRC that included a total of four products, including ZMapp and Remdesivir.
“This is the first-ever multi drug trial for Ebola. It has been implemented in a very difficult setting,” said Mike Ryan, executive director of the WHO’s health emergencies programme, at a press conference on 12 August.
‘Cure’ for Ebola?
REGN-EB3 and mAb114 show the best results, with mortality rates of 29 per cent and 34 per cent respectively, compared to 49 per cent for ZMapp and 53 per cent for Remdesivir.
"We're seeing even more remarkable results for people who receive early care," said Ryan.
In groups of patients who started treatment as soon as they were infected, mortality rates were around 6 per cent for REGN-EB3 and 11 per cent for mAb114.
Fauci stressed that he would not describe this as a “cure”, as some commentators have.“It is more appropriate to say that there is now scientifically sound data that have shown that two therapeutic interventions (REGN-EB3 and mAb114) can significantly decrease the mortality rate in patients with Ebola virus disease,” he said.
But he added: “It will give much hope to people with Ebola who previously had no effective treatment available to them.”