By: Sumita Thapar
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
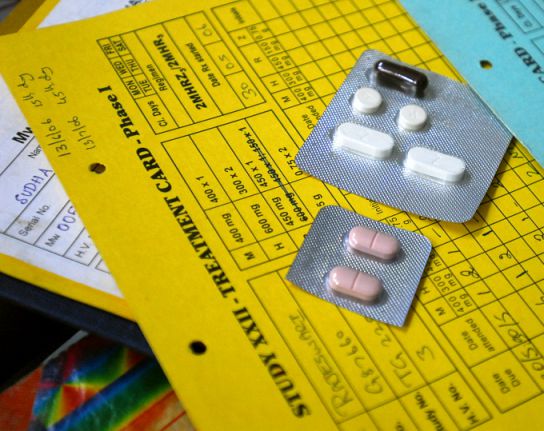
A tuberculosis drug specifically designed for children — combining sweet flavours and the correct dosage in a dissolvable tablet — is expected to hit markets early this year, according to the TB Alliance, a non-governmental organisation.
The tablets, which were developed through a partnership between the TB Alliance, the World Health Organization (WHO) and the United States government, are meant to improve drug regime adherence among children in the developing world. The drug is not new, but will be an improved combination of existing TB treatments such as rifampicin, isoniazid and pyrazinamide, specifically designed for children.
“Since the adult tablets were fixed drug combinations it meant redesigning the tablet according to the ratios [for children].”
James Seddon, Imperial College
At a conference in Cape Town, South Africa, in December, TB Alliance representatives told the audience that doctors and parents in many developing countries rely on crushing or splitting tablets with adult dosages when treating children with TB. This means drug dosage is difficult to control and many caregivers struggle to regularly administer the bitter-tasting, sharp-edged tablet fragments for the usual six-month course.
“Since the adult tablets were fixed drug combinations it meant redesigning the tablet according to the ratios [for children],” says James Seddon, a paediatric TB researcher at Imperial College, London in the United Kingdom.
Juliana Odindo, a Kenyan campaigner with Y+, a global network of young people with HIV, says the dissolvable drug is a huge improvement on her own treatments, which involved biting off small pieces of an adult tablet and swallowing them. “It is great news that child-friendly TB medication is finally being made available so that children do not have to gamble with their dosages like I did,” she says.
The WHO estimates that around one million children fell ill with TB in 2014, and 140,000 died from the disease. However, only around a third of those who catch the disease are officially diagnosed and put on treatment, the organisation warns.
According to Brenda Waning, head of global drugs at the Stop TB Partnership, drugs for children with TB make up a low-volume, low-profit market and are therefore not of interest for pharmaceutical companies. However, the partnership behind the child-friendly drug has pooled funding with UNITAID, an intergovernmental health initiative overseen by the WHO, to get the product to market.Manufactured by Indian pharmaceutical company Macleods, the drug is likely to be available early in 2016, the TB Alliance has said. Kenya, which has one of the highest burdens of TB, could be one of the first markets where the drug will be sold, according to Evaline Kibuchi, the TB manager at Kenya Aids NGOs Consortium.
“Our grassroots activists and civil society organisations have been advocating for child-friendly TB drugs over the last year,” she told SciDev.Net, adding that the only challenge remaining is approval by Kenya’s government.