03/04/26
Zimbabwe’s new HIV drug at risk as US funding retreats
By: Michael Gwarisa
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
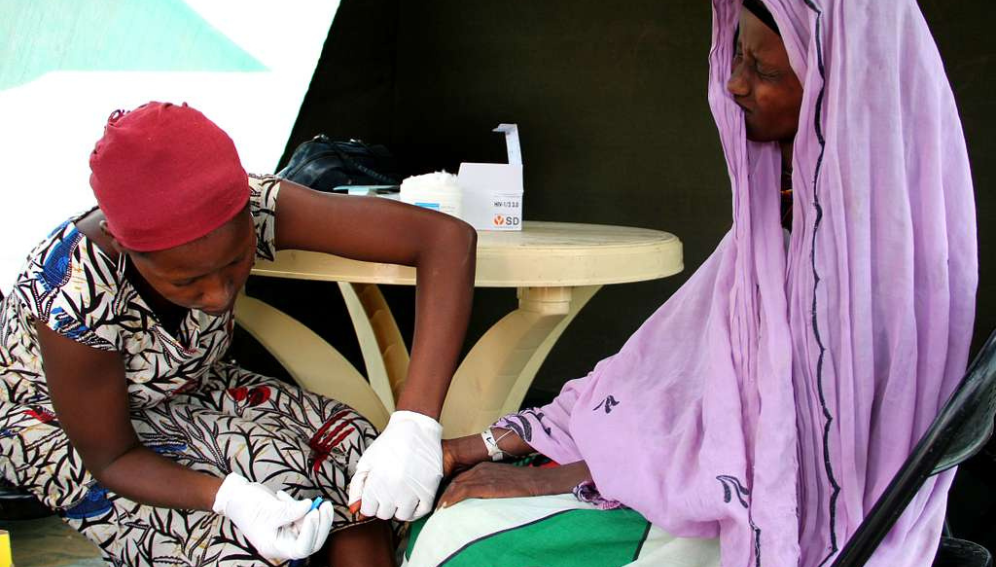
[HARARE, SciDev.Net] Nyasha Sayi recently received her first lenacapavir injection at a clinic in Epworth, a densely populated settlement on the outskirts of Harare. The twice-yearly HIV prevention shot felt like a lifeline—an escape from the difficult daily regimen of oral pills she relied on for protection as a sex worker.
“Daily oral prep pills were hard to maintain due to my work schedule,” the 31-year-old explained.
But with the United States pulling back health funding in Zimbabwe, she doesn’t know if she’ll get her second dose.
“As it stands, I am not sure if I will be able to get my next dose after 12 months,” Sayi told SciDev.Net.
“If it so happens that the programme is discontinued, it puts me back at risk.”
Sayi is among an estimated 2,000 people who received lenacapavir during a meeting with public health officials at the Zimbabwe Medical Association in Harare, which SciDev,Net attended on 31 January.
The drug is being introduced as part of a phase one rollout across 11 sites nationwide, targeting about 46,000 people at high risk of HIV infection, including sex workers, men who have sex with men, and other key populations.
However, that rollout is now clouded by uncertainty.
In February, US Ambassador to Zimbabwe Pamela Tremont, in a press statement shared with journalists, announced that would wind down major health interventions in the country, including programmes targeting HIV, tuberculosis, malaria, and maternal and neonatal health.
Zimbabwe has halted negotiations over a proposed US$300 million deal, according to a document from the Ministry of Foreign Affairs seen by this publication.
The negotiations broke down after the Zimbabwean government raised concerns that the agreement was extractive and imbalanced, alleging that the US government had sought access to sensitive pathogenic data and critical mineral resources.
The document directs Zimbabwe’s Ministry of Health and Child Care to discontinue talks and cites an off-the-record response from the United States Embassy in Zimbabwe, as well as a fact sheet outlining the terms of the agreement.
The US Embassy has denied these claims, maintaining it did not request access to sensistive data beyond standard data-sharing practices under programmes such as the President’s Emergency Plan for AIDS Relief (PEPFAR).
The withdrawal has left the future of lenacapavir—a long-acting injectable with near-perfect efficacy in preventing HIV—hanging in the balance.
Drug resistance risk
Scientists warn that delays to further doses could create a wider problem. According to Gilead’s prescribing information for lenacapavir (marketed in the US as Yeztugo), residual concentrations of the drug may remain in the body for up to 12 months or longer after the last injection, but at declining levels. If HIV exposure occurs when drug levels are no longer fully protective, there is a risk of developing resistance to this new class of prevention drugs.
Emily Gwavava, a consultant in Zimbabwe’s lenacapavir programme, said discontinuing treatment would put recipients’ lives directly at risk.
She told SciDev.Net: “Lenacapavir will remain in the body for another six months. So, after 12 months and sometimes even more, lenacapavir will still be there. But this will be a sub-protective level.
“The concern during that period is if clients continue to be at risk of HIV, and they contract HIV during that last six-month period, they may develop resistance to these capsid inhibitors.”
Those concerns are grounded in trial data presented at the 2026 Conference on Retroviruses and Opportunistic Infections, in Denver, US. In the Phase 3 PURPOSE 1 trial, a participant who missed a dose and discontinued lenacapavir contracted HIV 16 months after their last injection, the kind of gap in coverage that Zimbabwe’s funding uncertainty could produce.
Despite the alarm, Zimbabwean health authorities say current supplies are not under immediate threat.
Owen Mugurungi, director of the AIDS and tuberculosis unit at the Ministry of Health and Child Care, told SciDev.Net that existing doses were secured through a separate arrangement involving international agencies.
“The lenacapavir that is available is coming through a special arrangement involving the Global Fund, UNITAID and other partners, who negotiated pricing with Gilead,” he said. “The doses we currently have, have already been paid for.”
He added that the government had not yet received communication suggesting this supply would be disrupted.
Shifting sands
Zimbabwe’s situation comes as a number of African countries are navigating shifts in global health financing.
Yap Boum, deputy incident manager for Africa CDC, said the agency is closely watching how these financing arrangements are playing out across member states.
“We are observing how these arrangements are unfolding and what they mean for countries,” he said.
“Our role is to support member states to ensure effective implementation, accountability and transparency.”
Boum added that Africa CDC is also engaging countries to better understand challenges affecting participation in such agreements and to provide technical support where needed.
“These discussions are important because countries are at different stages, and the challenges they face are not the same,” he said.
For Sayi, that complexity means little. What matters is whether she will be back at this clinic in 12 months—and whether the injection she is counting on will be waiting for her.
This piece was produced by SciDev.Net’s Sub-Saharan Africa English desk.


