By: Wana Udobang and Emma Bryce
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
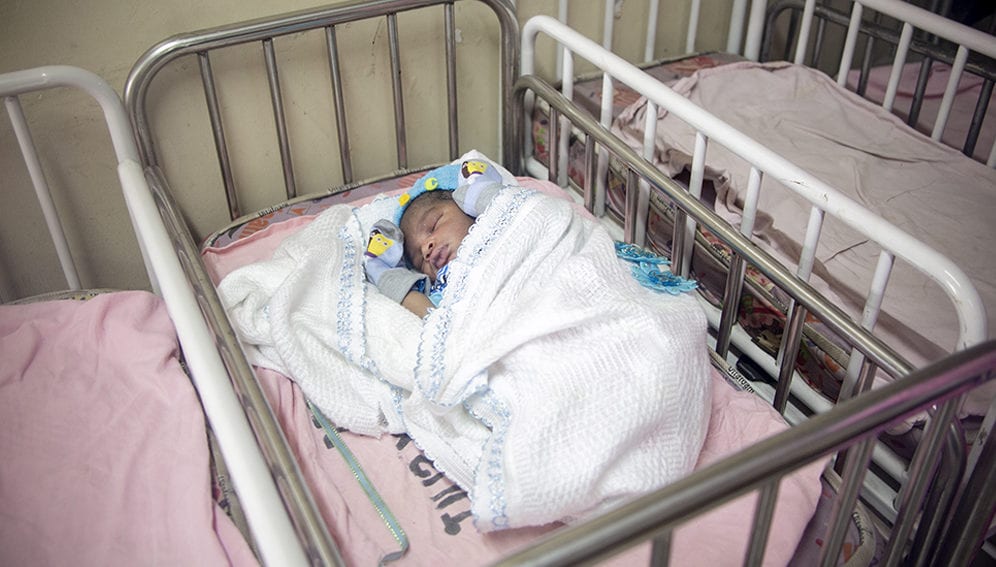
Peace Onuoha remembers the 6 April 2016 with conflicted emotions. It was the day she lost her daughter, and the day that doctors saved her life. She sits inside the mustard-coloured compound that is Nyanya General Hospital, on the outskirts of Abuja, Nigeria, and remembers how she was rushed here when blood began to saturate her clothes.
“They tried all day, but the bleeding refused to stop,” Onuoha says. “When they brought out the baby – my fair, beautiful baby – just like that, I lost it,” she says, quietly. Her child died shortly after birth. But as her bleeding worsened, the doctors gave her an emergency injection containing a drug called tranexamic acid, which slowed her haemorrhage, and Onuoha survived.
When Onuoha gave birth, tranexamic acid was being tested across Nigeria as part of the global WOMAN (World Maternal Antifibrinolytic) trial. The drug is already widely used to stem bleeding in trauma patients and during surgery. But this trial, launched in 2010 in 21 countries by researchers at the London School of Hygiene and Tropical Medicine (LSHTM), evaluated the effectiveness of the drug in childbirth, alongside other treatments for reducing postpartum haemorrhage.
“Excessive bleeding is killing people. I will not be quiet, I'm always telling others. We need to talk about it.”
Peace Onuoha
Nine years later, the results of the trial are in and there is good news for other women like Onuoha. It seems the drug is capable of saving many more lives. Nigeria in particular stands to benefit, because it has the second-highest rates of maternal mortality in the world. Forty thousand women die there every year during pregnancy and childbirth, and haemorrhaging is the largest cause of death, accounting for 23%.
But the legacy of the trial in the country is marked by both success and struggle: while some healthcare facilities record ongoing benefits to women, others are now dealing with dwindling supplies of the life-saving drug.
Globally, excessive bleeding after giving birth is a massive problem. It kills 100,000 women worldwide each year, making it the single biggest cause of maternal death. Tranexamic acid works by inhibiting the enzymes that usually break down blood clots, which slows bleeding. In 2017, the results of the six year trial showed that the drug prevented one-in-three deaths due to postpartum haemorrhaging. But timing is critical. “After every 15 minutes, you lose 10 per cent of the potential benefit,” says Haleema Shakur-Still at the LSHTM, who led the trial.
The drug proved so effective that the World Health Organization included tranexamic acid in its Essential Medicines List, a roster of the most crucial drugs hospitals should always have in stock.
Shakur-Still and Bukola Fawole, a doctor at the University of Ibadan who oversaw the trial in Nigeria, spoke to the country’s minister of health, Isaac Adewole, to try and ensure the drug’s future in Nigeria. Considering the large extent and success of the trial, they had a strong case: “As far as Nigeria is concerned, it probably has been the largest clinical trial conducted,” Fawole says..
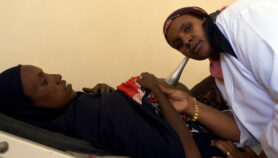
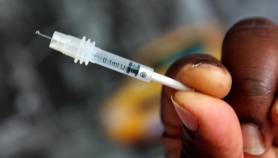
A mother tends to her newborn child at Ifako Ijaiye General Hospital in Lagos (Left); Two ampoules of tranexamic acid from the supply cupboard (Top right); Grace Adekola-Oni, a midwife at Ifako-Ijaiye General Hospital (bottom right).
Yet beyond issuing a nationwide directive to hospitals advising them to keep the drug in stock at all times, the government hasn’t taken any significant steps to bolster the drug’s supply. Adewole did not respond to SciDev.Net’s requests for comment.
Fawole says many hospitals aren’t even aware the drug exists. “Unfortunately it hasn’t been widely adopted, and that is an area where we need more work,” he says. “We must ask ourselves: what proportion of healthcare providers, doctors and midwives are aware of the findings of the trial? And then if they are aware, is the drug available to them?”
Haleema Shakur-Still says one doctor emailed her from a northern state in Nigeria to ask where he could obtain the drug. He was compelled to reach out because three women had recently come into his hospital at the same time with postpartum haemorrhage, and all three had subsequently bled to death. “He learned about the WOMAN Trial on Twitter, of all places,” says Shakur-Still.
But it’s not just a question of awareness. During the WOMAN trial, tranexamic acid was provided for free to participating clinics. But that’s over now. In Nigeria the drug costs between 1,000 and 1,550 Naira (£2.14 – £3.31) per ampoule. Each patient needs two ampoules – one gram in total – to ensure haemorrhage stops.
This is a cost that many resource-starved public hospitals shift onto their patients. But since more than half of Nigerians live on less than $2 a day, it’s too expensive for the majority of patients.
This struggle over pricing doesn’t occur everywhere. In Pakistan, the country that recruited the most patients for the WOMAN trial after Nigeria, tranexamic acid is now free at public hospitals, where one ampoule costs the government the equivalent of just 79 pence.
Whose responsibility?
There are plenty of essential drugs that end up being too expensive in Nigeria, which makes the case of tranexamic acid worth probing.
“It’s not a price controlled market in Nigeria. That is why everybody can put up a price tag of their own choice,” says Shaz Effendi, the manager of medical affairs at Hilton Pharma, a Pakistan-based company that manufactured tranexamic acid for Nigeria during the trial.
He says Hilton Pharma tried to keep the manufacturing price low; it was the distributor that applied the first increase in price on the drug.
This practice is common. A report published in 2011 by the United Nations Industrial Development Organization revealed that weak price controls in the Nigerian pharmaceutical sector enable mark-ups throughout the supply chain – from the manufacturer, to the distributor, wholesaler, and retailer – which can significantly increase the final price tag on a drug. In fact, a report published in 2006 by the Nigerian Federal Ministry of Health together with the World Health Organisation and others, found that drug prices in Nigeria were between 2 and 64 times higher than international reference prices.
According to Mojisola Christianah Adeyeye, director general of Nigeria’s National Agency for Food & Drug Administration & Control (NAFDAC), several companies now distribute the drug in Nigeria, including the pharmaceutical giant Pfizer, which also manufactures the drug. SciDev.Net contacted Pfizer for clarification about how they determine its price in Nigeria, but they declined to comment.
In any case, the system of perpetual markups means a drug’s price may change depending on where and when it is purchased, according to gynaecologist Hadiza Idris at Nyanya General Hospital. Even individual pharmacies can change the price seemingly on a whim. “When they notice you’re asking for something you don’t currently have in the hospital, of course they increase the price so they can make their own gain. That’s what they do around here,” Idris says.
If patients can’t afford to pay for the drug, her hospital still provides it, but then that eats into their emergency stock. “Most of the time when you give [the drug], that’s all, it’s gone. That’s one of the things hindering the continuous supply,” says Idris.
With all these players in mind, if the goal is to make the drug cheaper, then whose job is it? “That’s a million dollar question. Who is responsible?” says Shakur-Still.
Achieving affordability
Practically-speaking, Grace Adekola-Oni, a midwife at Ifako-Ijaiye General Hospital in Lagos, who was involved in the trial, reckons that even simply reducing the cost for her patients would make a measurable impact. “If it’s something like, say, 500 Naira, I think they could still afford that.”
Fawole hints that, in addition to price controls, the problem of affordability also boils down to an overarching health challenge in Nigeria: poor universal health coverage, which means that people accrue huge bills at hospitals that they can’t afford to pay all at once. “Very importantly we need to address the issue of out-of-pocket payment,” Fawole says
.In the meantime, a new spotlight is about to be placed on tranexamic acid. Currently the LSHTM is coordinating a second trial in Nigeria, Pakistan, Uganda and Zambia called WOMAN 2, to test the effectiveness of tranexamic acid in preventing haemorrhage in high-risk patients with anemia. This may help to underscore its potential for saving women’s lives.Back at Nyanya General Hospital, Peace Onuoha says that since the tragic loss of her daughter, she has since had another child–a son. She adds that her ordeal has driven her to raise awareness among others about the risks that women face in childbirth. “Excessive bleeding is killing people. I will not be quiet, I'm always telling others,” Onuoha says. “We need to talk about it.”
As for the nurses, midwives, and doctors on the ground, their hope is that one day, birth will no longer come with the spectre of death – an irony that overshadows the reproductive life of so many Nigerian women. “We are hoping that every woman will have the freedom to live, and that women will not be scared to carry a pregnancy,” says Aishatu Tenebe, a nurse at Nyanya General Hospital. “You know, it is sweet for a woman to come, deliver, and then go home.”
Reporting for this story was funded by the European Journalism Centre, through its Innovation in Development Reporting grants’ program.