By: Naomi Martin
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
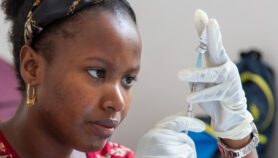
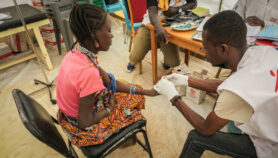
[LONDON] Total funding for research and development (R&D) on neglected diseases has suffered major cuts in the wake of the global financial crisis, although the impact has been reduced by a substantial increase in private sector funding, according to a major annual report.
The fourth Global Funding of Innovation for Neglected Diseases (G-FINDER) survey, launched today (8 December), found that year-on-year funding for neglected disease R&D decreased by 3.5 per cent (US$109 million) from 2009 to 2010 — the first overall decrease since the survey began in 2007.
The decrease resulted from lower contributions by the public sector, which still provided almost two-thirds of global funding in 2010, as well as by the philanthropic sector.
"It’s much worse than what we expected," Javier Guzman, director of research at Policy Cures, an independent non-profit research group based in Australia which published the report, told SciDev.Net at launch.
"Not so much because of the money itself, but because we saw cuts from [so many funders] — and we did not anticipate that."
Eight of the top 12 neglected disease funders have cut back their investment, Guzman said.
"We saw a very nice golden age, a very nice peak, but it was coming from very few people, very few aid agencies. Now these agencies are in trouble [and] we see the impact clearly," he added.
Research into diseases that rely on investment from public and philanthropic sectors — such as HIV/AIDS, malaria and diarrhoeal diseases — were hit the hardest, with funding for HIV/AIDS research decreasing by five per cent.
In contrast, diseases with substantial funding from industry, including tuberculosis and dengue, were largely protected.
Guzman predicted that the gap between the private and philanthropic sector funding would further narrow next year, but warned that the private sector would not be able to maintain a steady stream of funding on its own.
"The increasing private sector investments will still be there [next year]. But they are saying very clearly that partnership, co-funding and public support are needed," he added.
Guzman pointed out that most of the public sector funding is provided by the US National Institutes of Health, which concentrates on basic and ‘upstream’ research, rather than products in phase II and III clinical trials.
Even with the increased private sector funding, the shift towards basic research observed in last year’s G-FINDER report has continued, with product development partnerships suffering a funding decrease of US$47 million.
"We have a goldmine of knowledge and product portfolios," said Joris Vandeputte of the Tuberculosis Vaccine Initiative at the launch. "But these have to be funded to get them to the developing countries [that need them]".
Vandeputte said inventive, innovative funding mechanisms are needed to translate basic research results into affordable products.
Some possible avenues to follow include raising more money for this from emerging economies, or additional support from departments of trade and industry in the West, said Guzman.
"A wider base is important, with more people contributing, and more funders coming in," he added.
Link to full report ![]() [19MB]
[19MB]