By: Yojana Sharma
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
Science journalist Yojana Sharma finds the fight against counterfeits is deploying scanners, spectrometers, and minilabs — but nothing can replace national regulatory systems.
It is Accra, Ghana, and Samuel is in the pharmacy buying anti-malarial drugs for his fever. But he is worried: are they the real thing — or is he about to waste his cash on a few of the tens of millions of bogus drugs that percolate around developing countries, draining resources, risking lives and fostering drug resistance?
Help is at hand, however. Samuel finds a distinctive panel on the packet and scratches it to reveal a ten-digit code. He texts the code, for free, to a central number and a message comes back: fakes.
"If it is a ‘no’ message, the pharmacist must replace the drug — it is a criminal offence to sell fake medicines," says Bright Simons, president of mPedigree, the network he founded to fight the scourge of counterfeit drugs in developing countries.
mPedigree
Counterfeit drugs are a vast and growing problem for developing countries. Some 20 million fake and illegal medicines were seized in South-East Asia alone in 2010.
Bright Simons of the mPedigree – HP joint venture for drug authentication: "There is no profitable way for counterfeiters to beat the system".
mPedigree/BrightSimons
While most of the new technologies rely on analysing the make-up of tablets, this requires kit, and mPedigree neatly avoids this.
"It does not destroy the packaging or disrupt the security seal for the medicines," Simons says. An offshore data centre tracks the codes, which are randomly generated by the drug manufacturer. "There is no profitable way for counterfeiters to beat the system. You would have to go to a retailer and copy the codes, pack by pack. "
Simons is a Ghanaian rights activist-turned-social entrepreneur. He brought together manufacturers and mobile phone operators in Ghana, Kenya and Nigeria to implement mPedigree, and roll-outs are planned for Uganda and Tanzania, with India and South-East Asia possibly joining later. Simons’ work has earned him a Grand Prix Net Explorateur award, an international prize for the world’s best digital innovation.
The key, he says, has been gaining trust from pharmaceutical manufacturers and telecommunications operators.
All the technology infrastructure is in Europe which Simons says makes it easier for manufacturers to trust the system.
Mobile phone coverage is still not perfect — to date 80 per cent of telecommunications providers in the three countries participate. But it works well enough for the Nigerian government to have incorporated mPedigree into its national quality assurance standards for pharmaceuticals.
A key problem now is awareness-raising. "It will be a while before it is a household service," Simons admits.
Simons acknowledges that mPedigree cannot replace the laboratory testing underpinning most attempts to tackle counterfeit medicines. But, he contends: "You will need fewer mobile laboratories for testing and the laboratories can concentrate on particular points of entry such as the ports".
A lab in a van
Getting laboratory facilities to where they are needed is crucial for counterfeit drug detection. In one much-admired initiative, researchers at China’s National Institute for the Control of Pharmaceutical and Biological Products equipped vans with spectrometers, which analyse the spectrum a compound emits when exposed to near infra-red (NIR) light. The vehicles also carry an arsenal of other test kits including thin-layer chromatography and colorimeters.



Scratch the panel and discover if your medicine is genuine
mPedigree/BrightSimons
Some 400, well-equipped vans now travel through the Chinese countryside testing medicines.
"In China, as everywhere in the world, counterfeit medicines and substandard drugs are mainly in rural areas where drug administration is weak," says Jin Shaohong, executive deputy director of the institute, in an online forum.
His group is now developing van minilabs that also offer High Performance Liquid Chromatography (HPLC). The advantage, says Jin, is that "if a drug is suspected by NIR screening then an immediate on-site confirmation can be performed by HPLC".
Previously, vans in rural areas had to refer suspicious drugs from local clinics and pharmacies to central laboratories.
"These mobile mini labs are technologically very good," says Lembit Rägo, the WHO’s coordinator for quality assurance and medicines safety.
"They can travel in the country and keep some sort of detection net in place.
"But the pre-condition is that you have to have one original drug in your possession to compare the spectrum," he adds. With a burgeoning manufacturing industry, particularly for generic medicines, this may not always be possible.
The vans do carry a library of NIR spectra for pharmaceutical products available in China. But Jin accepts that more spectra are necessary because new formulations regularly arrive on the market. Because of this, China is setting up a national spectrum database.
Marked medicines
The Chinese system is relatively expensive. Most developing countries check visually for counterfeits, with customs officials or pharmacists looking for non-standard packaging. But it is a game of spiralling sophistication on both sides. Counterfeiters are becoming particularly adept at creating holograms for packaging.
Some pharmaceutical manufacturers mark not the packaging, but the actual tablets, making the marks hard to detect and copy. Others add radio-frequency tags to packaging, so that the batches can be tracked. Both methods make essential medicines more costly — a problem for resource-poor countries — while not always preventing counterfeits. And suspect marked or tagged drugs must still be analysed: the portability of these analysis systems is key.
Paul Newton, an infectious diseases doctor at Mahosot Hospital in Laos, uses colorimetric kits, developed with Michael Green at the Centers for Disease Control and Prevention in Atlanta, United States, and popularly known as the ‘dye test’.
"It is not really a dye, it’s a chemical that reacts with the active ingredients to produce a yellow colour (in the case of antimalarials). You can judge the quality of the medicines according to the colour intensity," says Green.
Another colorimetric test requires more technology but less training. "You take a picture using a digital camera or mobile phone, and free software downloadable from the internet can measure the colour frequency distribution," says Green. Colorimetric approaches are widely used in Cambodia, Laos and Thailand, and by non-governmental organisations and hospitals to screen anti-malarial drugs in Africa and Asia. They are becoming a standard part of minilabs.
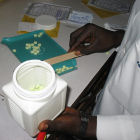


Drugs: to trace the fraud properly you need a fully fledged lab
Flickr/MikeBlyth
The most widespread minilab is that of the Global Pharma Health Fund (GPHF), with almost 400 in 70 countries, mainly Africa and Asia and the Western Pacific. Combining colour-testing and chromatography, it packs into two very large suitcases and costs US$10,000 (GPHF subsidises this by US$5,000 and throws in the US$2,000 reference drugs for free). Consumables cost up to US$4 per test.
This is cheaper than a spectroscopy lab, which can cost US$40,000 alone for the initial kit. up front plus for the kit alone. Each spectroscopy test also costs another US$4 in solvents and other consumables (often flammable and hard to transport). In most countries they are not transported around but remain in capitals or provincial cities.
Each analysis, which generates a result in minutes, costs a fraction of the normal chromatography-based tests and does not need reference materials to compare the results, Rohrbasser says.
Mali’s national laboratories in Bamako are already using a Pharmelp minilab, and Cambodia’s National Health Product Quality Control Centre in Phnom Penh uses another. There are plans to manufacture enough units to send to Congo and, by the end of 2011, to Burkina Faso, Madagascar and Senegal.
Another minilab that does away with expensive consumables and solvents but remains portable, is being developed at King’s College London, in the United Kingdom, together with scientists from Lund University in Sweden. Based on quadrupole resonance (QR) spectroscopy, akin to magnetic resonance imaging, "it is non-destructive, non invasive. You put the packet into the system unopened, then return it to the shelf or give it to the patient," says Jamie Barras, a research associate at King’s College London.
Although far from field-ready, the lab will "unambiguously identify over 10,000 different chemical substances," Barras says. And it does not need high-level scientific training to use.
Hand-held digital spectrometers from Thermo Fisher, in use in Nigeria, are another attempt to meet the portability criterion. "The great hope at the moment is gun-like instruments that scan through the pack," says Newton.
Regulation still required
But portability is also a limitation. "At the end of the day you will need a complete elemental analysis at a fully fledged lab and to relate that to previous data held (on that drug)," says Barras.
That is because regulators need to understand what is being substituted for the real drugs, and what is being left out, and to try to identify the manufacturer.
"I do not foresee a time when anyone [such as a medicines regulation agency] would rely only on what is gathered in the field," says Barras. Minilabs can certainly save lives by detecting the worst contaminations and infringements. But even the best "can be up to one third less sensitive compared with full quality control," says the WHO’s Rägo.
"Counterfeiters are very smart. They may put a small amount of active ingredient in the fake so that it can pass minilab tests." And these are all "after-the-event technologies" that do not prevent counterfeiting, or their huge financial costs to poor countries.
"You have to be able to track along the whole supply chain," Rägo says. "There is no single answer. Minilabs have a role in the field but they can’t be everywhere and test every pack. It is too costly. There is no substitute for a fully functioning regulatory system."
Yojana Sharma is a freelance science journalist.
This article is part of a Spotlight on Detecting counterfeit drugs.
In response, academics, regulators and industry are rolling out new technologies to try and get a step ahead of the counterfeiters. But even the technologies’ inventors admit that nothing can replace a properly functioning national pharmaceutical regulatory system.













