Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
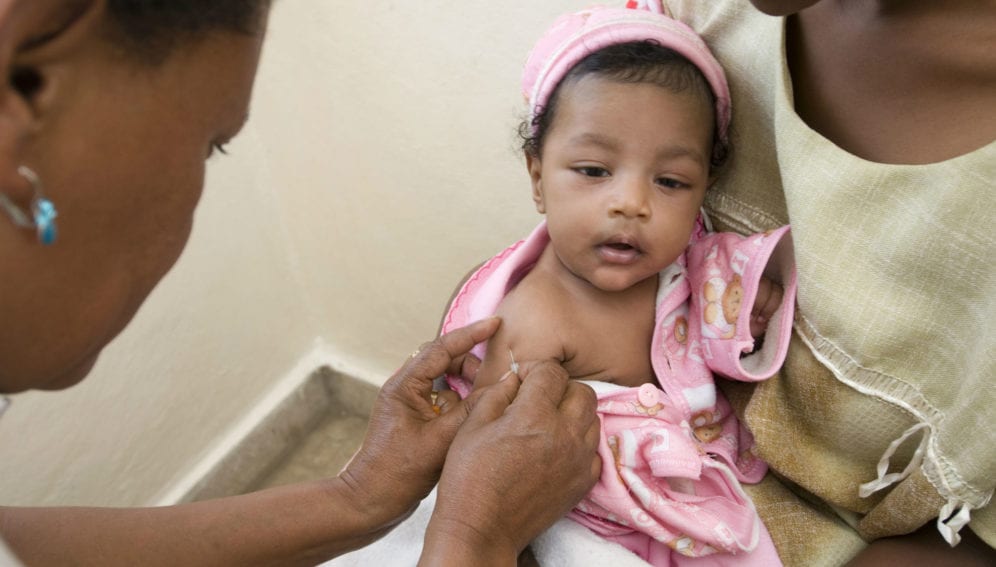
A single Bacille Calmette-Guerin (BCG) dose given to adolescents who had been vaccinated as infants could prevent a sustained tuberculosis (TB) infection by 45 per cent, a study suggests.
The results were presented at the 5th Global Forum on TB vaccines in New Delhi, India last month (19 February).
The World Health Organization (WHO) says that about a third of the world's population has latent TB infection — people infected by TB bacteria but are not ill with the disease and cannot transmit the disease.
“The robustness of the BCG results to help clear or control an infection was surprising”
Ann Ginsberg, Aeras
The clinical trial was carried out at the South African Tuberculosis Vaccine Initiative (SATVI) and the Emavundleni Research Centre at the Desmond Tutu HIV Centre in the Western Cape of South Africa to assess TB prevention with vaccines.
Researchers randomly assigned 990 HIV-negative, healthy adolescents aged 12 to 17 years of age, vaccinated with BCG as infants to receive one of three vaccines: a placebo, H4:IC31—an experimental vaccine— or BCG, a TB vaccine in existence for close to a century. The trial began in March 2014.
The trial measured the rate at which individuals tested positive to TB infection at six months intervals over a period of two years.
According to the findings, the vaccine efficacy for preventing a sustained infection was about 45 per cent whereas that of the experimental vaccine was about 30 per cent.
Mark Hatherill, director of the SATVI at the University of Cape Town, and the study's principal investigator, said in a statement that the first-known prevention-of-infection trial for TB showed that vaccination has the potential to reduce the rate of sustained TB infection in a high-transmission setting.
“The robustness of the BCG results to help clear or control an infection were surprising,” said Ann Ginsberg, chief medical officer for Aeras, an nonprofit organisation advancing the development of new tuberculosis vaccines
BCG showed efficacy in preventing sustained TB infections as participants who initially tested positive for TB infection were more likely to have cleared or controlled the infection within six months.
The experimental vaccine showed some ability to prevent sustained TB infections but not at statistically significant levels, she said.
“This study provided valuable, modern data on a very old vaccine – data that the field can use to potentially optimise BCG vaccination strategies and, create new, more effective TB vaccines,’’ Ginsberg told SciDev.Net, adding that the study did not assess the use of BCG to prevent TB disease and that additional studies are needed.
“This is a promising trial. However, the evidence on the tested vaccine strategies is insufficient,” added Claudia Denkinger, head of TB programme at the Switzerland-based FIND, an international non-profit organisation for development and delivery of much-needed diagnostic tests for poverty-related diseases.
Testing after six months showcases lack of sufficient number of diagnostic tests to prove the efficacy of the vaccine, Denkinger said.“This might be in part due to the fact that diagnostic tests available to such vaccine trials are insufficient for TB. For other diseases such as hepatitis B and measles, we have diagnostic tests that clearly indicate infection or protection from disease. In TB, we don’t,” she told SciDev.Net. The WHO has identified the need for better tests for TB and has drafted product specifications.
“Without a concerted effort on vaccines and diagnostics, we will not be able to achieve the WHO End TB targets,” Denkinger added.
This piece was produced by SciDev.Net’s Sub-Saharan Africa English desk.