18/01/21
Supply delays hit planned Ebola vaccine stockpile
By: Ruth Douglas and Julien Chongwang
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
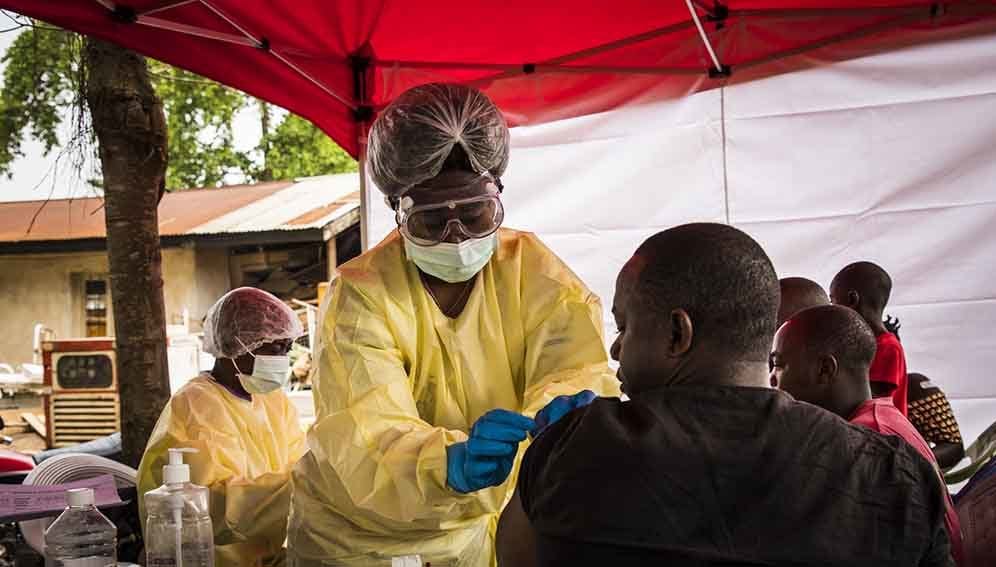
Supply chain delays must be addressed to speed up creation of a stockpile of vaccines against deadly Ebola disease, researchers say.
Once it is fully operational, low and lower middle-income countries will be able to access the stockpile of 500,000 doses free of charge along with support for operational costs for the rollout of immunisation programmes, the vaccine alliance Gavi and partners announced Tuesday.
However, international organisations warned it could take up to three years to reach the target.
“By creating a stockpile of 500,000 doses of the Ebola vaccine, available to all countries, we can help prevent loss of life and swiftly end Ebola outbreaks in the future,”
Gavi CEO Seth Berkley
The new Ebola vaccine was used to tackle a two-year long outbreak in the eastern Democratic Republic of Congo, which was declared over last June after 300,000 people were immunised.
The injectable Ervebo vaccine (rVSV-ZEBOV-GP), manufactured by Merck, Sharp & Dohme (MSD) Corp., is effective against the most common Zaire strain of the virus. It has been approved by eight African countries following a fast-tracked prequalification process by the World Health Organization (WHO), certifying its safety and efficacy.
The stockpile will include licensed doses of Ervebo, which has also been approved by European and US regulators, said Gavi, as well as other candidate vaccines in the pipeline once licensed.
The stockpile will be housed in Basel, Switzerland, and managed by an international coordinating organisation, comprising UN agencies the WHO and UNICEF, and aid agencies Doctors Without Borders (MSF) and the International Federation of Red Cross and Red Crescent Societies (IFRC).
On request from a country, vaccines can be made available together with ultra-cold chain packaging by the manufacturer for emergency response shipment, the WHO said.
However, MSF warned that there were currently only 6,890 doses available because of “supply bottlenecks” and said it could take up to three years for the 500,000 target to be met. The WHO also said it could take two to three years.
David Heymann, of the London School of Hygiene and Tropical Medicine, said: “The challenge for the Ebola stockpile is to have enough vaccine immediately ready to roll out – hopefully there will be more vaccines to add to the stockpile in the future.”
He stressed that the vaccine was only licensed for the Zaire Ebola strain and may not work effectively for other strains.
The first deliveries of doses into the stockpile are being funded through a US$20 million contribution from the United States Agency for International Development (USAID).
Since Ebola outbreaks are relatively rare and unpredictable, there is no natural market for the vaccine, the WHO explained.
Gavi said it had created “strong incentives” for vaccine manufacturers through an advance purchasing commitment to procure doses once they were prequalified by the WHO.
Development of the Ebola vaccine was expedited following the 2013-2016 epidemic in West Africa, which left more than 11,300 dead in Guinea, Liberia and Sierra Leone.
Guyguy Manangama, head of the Ebola response at MSF, who worked on the last Ebola outbreak in the DRC, said vaccination was one of the most effective ways of fighting Ebola epidemics and the stockpile would allow rapid deployment in endemic areas, but added it “should not limit efforts for the development of other vaccine candidates”.
“It will be necessary to ensure a good system of replenishment of this stock so as not to deal with disruptions on the ground at critical times of epidemics, to continue research in order to improve and adapt the cold chain according to the context, so that to have a more stable vaccine that does not require a complex cold chain,” he said.
COVID-19
Gavi chief executive Seth Berkley said the Ebola vaccine had “set a precedent for fast-tracking development and production of vaccines against COVID-19”.
Gavi is also jointly leading the COVAX programme with the WHO, aimed at procuring and ensuring equitable distribution of COVID-19 vaccines.
“By creating a stockpile of 500,000 doses of the Ebola vaccine, available to all countries, we can help prevent loss of life and swiftly end Ebola outbreaks in the future,” Berkley added.
Marianne Comparet, director of the London-based International Society for Neglected Tropical Diseases, was optimistic, despite the time it will take to reach the full stockpile.
“The fact that such major organisations have partnered together on this will ensure accelerated scrutiny, at the highest level and in-field, of the supply-chain bottlenecks,” she said.
A spokesman for MSD said the company was working closely with the WHO on replenishment and supply of the vaccine.
“We are moving swiftly to manufacture licensed doses of Ervebo, and this can take appropriate time. In the interim, we have been committed to continuing to provide investigational doses for outbreak response,” he said.
This article was originally produced by SciDev.Net Global edition.















