26/11/20
Asian countries need help to access COVID-19 shots
By: Sanjeet Bagcchi
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
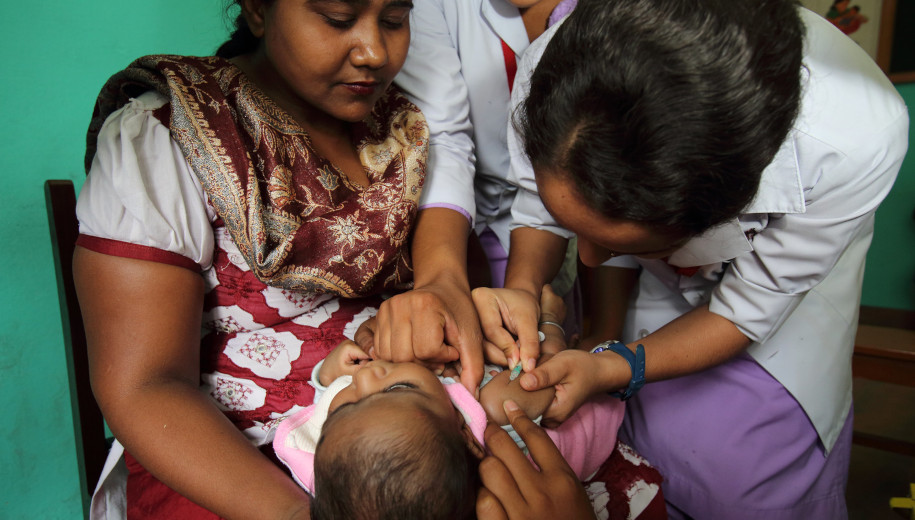
Most Asian countries will need support to equitably access COVID-19 vaccines.
[NEW DELHI] As three pharma majors, Pfizer Inc with partner BioNTech SE, Moderna Inc. and AstraZeneca Plc announced this month the effectiveness of their experimental vaccines in preventing COVID-19, there were no guarantees that developing countries will get their fair share.
However, there was hope from the Asian Development Bank (ADB) and other partner groups that some measure of funding support would be available to its member countries. In April, ADB had approved measures worth US$20 billion to streamline quicker and more flexible delivery of assistance against the COVID-19 pandemic. More recently, it allocated another US$20.3 million for technical assistance.
The new allocations will be available for ADB developing members to “support vaccine-related health system assessments and the development of country readiness plans to strengthen the capacity to access, introduce, deploy, deliver, and monitor vaccines safely and effectively”, an ADB press release dated 17 November said.
“For a number of developing countries in the Asia and Pacific region, support is essential to secure and distribute the vaccine early”
Anish Ray, Cook Children’s Medical Center
Woochong Um, director-general of ADB’s sustainable development and climate change department, said in the statement that “Asia Pacific has largely done well to limit the spread of COVID-19. Ensuring access to a safe, effective, and equitable vaccine is the next frontier in the fight against this virus”.
COVAX (short for COVID-19 Vaccines Global Access), an initiative launched by WHO, the vaccine alliance Gavi and the Coalition of Epidemic Preparedness Innovations, is expected to help developing countries, including those in Asia, secure vaccines early to fight the pandemic.
Anish Ray, a paediatrician at the Cook Children’s Medical Center, Texas, US, says that countries like China, Japan, Singapore and South Korea may secure a vaccine early owing to their financial, infrastructural and technological strengths.
“But for a number of developing countries in the Asia and Pacific region, support is essential to secure and distribute the vaccine early,” he tells SciDev.Net.
With more than 180 member-countries, COVAX — an initiative worth US$18 billion — aims to have two billion doses of vaccine available by the end of 2021, which should be enough to protect high-risk and vulnerable people, as well as frontline healthcare workers, according to Gavi.

Funding inadequacy
In a 13 November statement, Gavi said that with 94 higher-income economies joining the alliance, COVAX can support 92 countries with low-and middle-income economies in gaining access to safe and effective vaccines for COVID-19.
However, an editorial published on 21 November in The Lancet notes that “COVAX, the Gavi-led financing mechanism to provide COVID vaccines to low-income and middle-income countries, has raised US$2 billion, but needs US$5 billion more for 2021”.
According to Diptendra Sarkar, a COVID strategist, public health analyst and professor at the Institute of Postgraduate Medical Education and Research, Kolkata, India, “It is important that authorities in various developing countries of the Asia Pacific region — such as Nepal, North Korea, Indonesia, Myanmar, the Philippines, Sri Lanka, Timor-Leste and Vietnam — stay cautious with respect to securing the COVID-19 vaccine in time as per their specific requirements, in coordination with COVAX, a busy international facility.”
Sarkar tells SciDev.Net that developing countries need to prepare a priority list according to which the vaccine would be secured and distributed. In India, for example, the vaccine will be given to the health care workers first, then to older people and those with co-morbidities. Similarly, in countries like Malaysia, the vaccine will be first given to doctors, nurses, police and other frontline workers.
Leading the COVID-19 vaccine race is ChAdOx1 nCoV-19 — developed by the University of Oxford, pharmaceutical company AstraZeneca. It has collaborators, such as the Indian vaccine manufacturer Serum Institute of India, which expects to launch the vaccine, called COVISHIELD, within a few months.
India is also developing indigenously Covaxin, developed by Bharat Biotech and its collaborators. This vaccine is currently on phase 3 trials involving 26,000 participants across 26 centres in the country.
In August 2020, Gavi, Serum Institute of India and the Bill & Melinda Gates Foundation formed a collaboration to provide 100 million doses of their candidate vaccine to low- and middle-income countries through the COVAX facility, at US$3 per dose.
However, according to The Lancet editorial, Pfizer and Moderna – two other key vaccine developers from the West – have not yet reached agreements with COVAX to supply vaccines. “Although some vaccine developers have promised to limit profits from the COVID-19 pandemic, Pfizer and Moderna have made no such commitments,” the editorial said.
According to Sarkar, within the COVAX facility, nine candidate vaccines are being assessed through various stages of clinical trials; China and the US have two vaccines each, South Korea and the UK have one vaccine each, and one vaccine belongs to the global partnership.
India an exception
For developing countries in South Asia and South-East Asia, the impact of rich, developed countries in terms of COVID-19 vaccine’s supply and distribution may be significant, despite the facilities and services provided by ADB, COVAX and other global bodies. The exception is India.
“Unlike India, not all the developing countries in the Asia Pacific region are self-sufficient to develop their own required vaccine during the pandemic,” explains Sarkar. “Most of the developing countries may have to depend on some other countries in securing the vaccine, and thus an atmosphere of mutual understanding and collaboration will become significant in days to come,” he adds.
“South Asian and South-East Asian countries will have to make hard choices with major geopolitical implications,” says Lawrence Gostin, professor of global health law at Georgetown University in the US and director of the O’Neill Institute for National and Global Health Law.
According to Gostin, India is the most important of all the low- and middle-income countries. “It has a vast pharmaceutical industry that is technically capable of manufacturing vaccines in high volume. India could use its strategic advantage not just for its own needs but could also be a major exporter of vaccines,” he tells SciDev.Net.
With COVID-19 affecting 60.72 million people globally and killing 1.426 million (as of 25 November), scientists, health professionals, international organisations and the general public — are all awaiting a vaccine.
Thekkekara Jacob John, emeritus professor and former head of clinical virology at the Christian Medical College, Vellore, India, tells SciDev.Net: “We need the vaccine now, not after the epidemic dies down naturally and transitions into endemic phase. Protection delayed is protection denied.”
This piece was produced by SciDev.Net’s Asia & Pacific desk.