Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
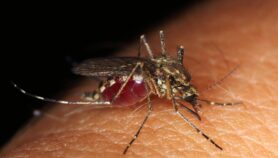
[DURBAN] The global malaria medicine chest could be boosted with a new drug researched and developed in Africa.
MMV390048, a drug compound developed in Africa to combat malaria, is expected to go on phase I clinical trials within the first six months of next year, delegates attending the 6th Multilateral Initiative on Malaria Pan-African malaria conference in Durban, South Africa, were told early this month (8 October).
The phase I trial in about 20 to 80 healthy volunteers will take place at the University of Cape Town's (UCT's) Groote Schuur Hospital.
The next-generation antimalarial compound, MMV390048, underwent a year of pre-clinical development after discovery in 2012 at the Drug Discovery and Development Centre (H3-D) at the UCT in South Africa.
International partners for the project include Switzerland-based Medicines for Malaria Venture, Monash University in Australia and Swiss Tropical and Public Health Institute in Switzerland.
"The journey of a compound from bench to bed is long," Kelly Chibale, founder and director of H3-D tells SciDev.Net.
Success in this phase means the drug will go through two other phases before it can be submitted to a stringent regulatory authority for World Health Organisation pre-qualification, Chibale explains. The process takes at least six to eight years, assuming that the compound passes each clinical development phase.
The new drug could reduce the malaria burden of 600,000 deaths worldwide each year, with 80 per cent being children and 91 per cent occurring in Africa.
The focus is now on research into new medicines for malaria since many of the old drugs relied upon to cure malaria have fallen to resistance, says Chibale.
Globally, malaria drugs face such challenges as low quality, drug resistance, affordability and non-compliance.
“Due to its potential as a single dose cure, MMV390048 would improve patient compliance, as taking one pill would be far simpler for patients”
Kelly Chibale, H3-D
Due to its potential as a single dose cure, such characteristic of the drug is unlike any of the current medicines in the studies done so far, says Chibale
Solomon Nwaka, acting director of African Network for Drugs and Diagnostics Innovation, says the clinical trials should not be a cause for unease as the studies will be well planned and executed by medical experts.
"I understand the concern that sometimes drugs and vaccines trials are conducted in Africa, but when the product reaches the market, Africans cannot fully access them due to high cost and other access issues," adds Nwaka. "Hopefully this situation will change for good."
This article has been produced by SciDev.Net's Sub-Saharan Africa desk.