By: Gita Ramjee
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
As women have come to represent the face of HIV/AIDS in Africa, so interest has grown in microbicides as a ‘woman-controlled’ option to prevent HIV infection. Microbicides are products formulated as gels, creams or suppositories that can be applied to the vagina or rectum before sex.
In the early 1990s, I worked on a small study of the acceptability of microbicides to sex workers. Then, in the South African Medical Research Council’s HIV Prevention Unit, I contributed to the Col-1492 trial, a phase 3 study of a vaginal microbicide, nonoxynol-9. Although it had a negative outcome — the product increased the risk of HIV infection — the HIV Prevention Unit and the South African MRC became known for clinical trials of microbicides.
A growing field
With our insight into ethical challenges in conducting clinical trials in developing countries, the HIV Prevention Unit is now involved in four microbicide trials and one vaginal diaphragm trial for the prevention of HIV in women.
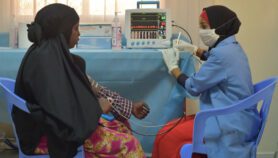
.jpg) |
| Women reviewing information during an HIV/AIDS awareness training for teachers Credit:USAID |
Everyone benefits
Working in Kwazulu Natal, the area with the highest prevalence of HIV in South Africa, we select populations in which research has never been done, in different geographical locations chosen to avoid competition for participants and to avoid communities receiving mixed messages from different research groups.
Every community in which we conduct trials benefits from our comprehensive HIV prevention efforts — education programmes, lay-counselling courses, and training on home-based care within the community. These services are provided irrespective of trial participation or whether the tested intervention is proven effective.
Not all research groups provide this package of benefits. However, we have learnt that effective community participation occurs when researchers are prepared to give something back to the communities in which they conduct trials. We should empower communities to look after themselves by implementing sustainable HIV prevention programmes, regardless of trial outcome.
As soon as we begin planning the protocol for a trial, we meet with community stakeholders to discuss any objections or concerns they might have so that we can resolve these problems before implementing the protocol.
One of the key ethical issues we need to address is that of care, especially for those who become HIV-positive during the trial. We are working with our sponsors to build the capacity of local health providers so that they can offer care for 3–5 years after the trial ends. As researchers, we have an ethical obligation to facilitate, although not necessarily provide, care for the participants who become HIV-positive.
Our trial participants volunteer mainly because our education and care programmes make them aware of their health status and because they want to remain HIV-negative.
Attitudes to microbicides
Men are supportive of microbicides, but we have seen some resistance to the label ‘woman-controlled’ because the term ‘controlled’ has negative connotations for men within the context of a partnership. Instead, they would like these products to be called ‘woman-initiated’.
There is a risk that the use of microbicides will encourage couples to abandon the use of condoms — so-called ‘condom migration’ — especially in sub-Saharan Africa where there is a dislike of condoms. To counteract this potential problem, especially with the first generation of microbicides, which may not be more than 33 per cent effective, the product will need to carry a label recommending that it be used in combination with condoms. We would rather encourage people to use dual protection such as condoms and microbicides, so that if they do have sex without a condom, the protective effect of the microbicide, albeit weak, will still have an impact on HIV prevention.
We do a lot of education to prevent misunderstandings and stigmatisation of trial volunteers. For example, we try to dispel rumours that the trial is for HIV-positive people. We spread the message that the trial is for HIV prevention, and any diagnosis of HIV infection is kept confidential. Most of the communities that we work in are very supportive. I personally give feedback on our data to the community every six months. Communication is essential for a successful trial; you cannot go into a community and work in isolation.
Lessons for vaccine researchers
Testing in humans is more advanced for microbicides than for vaccines, and I believe that vaccine researchers could learn from the experiences of scientists working on microbicide trials.
First, both groups could work together on developing a community care package that includes training and building capacity for local service providers to facilitate care. This way, vaccine trialists could avoid having to address these issues at a later time when they implement vaccine trials in the same country, region, or community.
Second, if vaccine researchers work with established phase 3 trial sites in developing countries, they can make use of the same infrastructure and community partnerships set up by microbicides research groups.
I have not been approached by anybody from the vaccine community and I am surprised at this short-sightedness. Our goal — HIV prevention — is the same, irrespective of the intervention. With this lack of collaboration, I believe we have missed opportunities to have common trial systems in place, to establish a site for a phase 3 vaccine trial, and to capitalise on existing effective strategies and community partnerships.
Interview by Julie Clayton
Gita Ramjee is Director of the South African Medical Research Council’s HIV Prevention Unit in Durban.
Read more about microbicides in SciDev.Net’s microbicides spotlight.