By: Evelyn Harvey
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
Controlling tuberculosis in the developing world means investing in suitably equipped labs as well as in diagnostics, argues Evelyn Harvey.
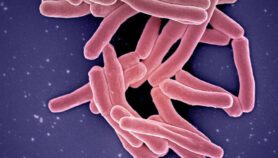
Tuberculosis (TB) is a stubborn and deadly disease, difficult both to detect and treat. Arguably the world’s most successful pathogen, Mycobacterium tuberculosis causes 1.6 million deaths from TB every year and latently infects one in three people.
Extensively drug-resistant strains (XDR-TB) and co-infection with HIV are undermining the progress made in curing TB since the first drugs became available in the 1940s.
But even new drugs designed to address these problems will not hold the balance in TB control. Without an accompanying improvement in TB diagnostics, we shall quickly reach a new impasse.
Tuberculosis often goes undetected in developing countries, and people seeking treatment are not tested for drug sensitivity — a strategy fundamental to the control and treatment of drug-resistant TB — mainly because of limited resources.
These countries urgently need a quick, cheap and technically straightforward diagnostic test for TB and a rapid means of identifying drug-resistant strains to prevent outbreaks of XDR-TB.
Accomplishing these aims calls for more than simply increasing funding for research into TB diagnostics. It will also require a commitment in the countries with the highest TB burdens to increase the number of suitably equipped laboratories in the field and to laboratory training.
Advances in diagnostics and drug-sensitivity testing will also need to be technologically accessible and sustainable in the often remote settings in which they are likely to be used.
Diagnostics in the pipeline
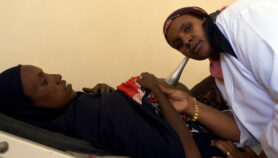
Sputum microscopy — where slides of sputum from a patient’s chest are stained for the TB mycobacterium — has long been the gold standard of TB diagnosis. It is still implemented under DOTS, the internationally recommended strategy of the World Health Organization (WHO) for TB control.
But it has severe limitations in the developing world. Diagnostic accuracy in HIV-positive individuals is dismal, and it can take weeks to obtain a diagnosis, causing considerable loss of income and travel costs for the patient.
There are already broad moves underway to improve TB diagnosis. In 2003, for example, the Bill and Melinda Gates Foundation made US$30 million available for research into TB diagnostics.
Likewise, the TB Diagnostics Initiative run by the WHO is working with multiple industrial and funding organisations to develop new diagnostic tests for use in low-income countries.
Another initiative is the Foundation for Innovative New Diagnostics (FIND), which is working with nearly 40 companies, academic institutions and government bodies to develop simple TB tests that can be used on the spot.
They have several new diagnostic tools in the pipeline, including culture-based tests to identify TB and determine any drug resistance, and assays to detect antigens or DNA from the TB pathogen and the patient’s immune response to it.
But although such tools can be very effective at rapidly detecting TB infections, many are prone to invalidating contamination or require technical expertise or specialised equipment that may not be available in resource-poor settings.
Improving laboratory facilities
Some culture-based and drug-sensitivity tests — for example, nitrate reductase assay or microcolony analysis — can be carried out using unpatented laboratory techniques that do not require the purchase of kits, equipment or reagents.
But very little is being done by governments and donors to develop laboratory facilities in countries with a high TB burden. This neglect is short-sighted as it has left many regions incapable of performing drug-resistance testing or culture-based diagnosis.
Any call to invest more in the research and development of new diagnostic tools for TB must be accompanied by an appeal for increased spending to improve basic in-country laboratory infrastructure and training.
There is little to be gained from attempting to implement high-tech, high-maintenance and high-cost procedures in resource-poor settings. Yet last year, when Médecins Sans Frontières approached several companies who were developing TB diagnostics with a view to analysing those in the pipeline, the organisation found that very few companies were aware of the reality of the poor working conditions and limited resources available on the ground.
Techniques developed in affected countries will be less removed from the reality of TB control than those emerging from well-funded and well-equipped labs abroad. Capacity building for TB research and development in resource-poor countries must be seen as a global priority.
The WHO and Stop-TB partnership aims to halve TB prevalence worldwide by 2050. Our best chance of achieving this ambitious target is to combine new diagnostics research with efforts to scale up the methods already available and implement them where they are most needed — in the peripheral outposts on the frontlines of the TB emergency.
Evelyn Harvey is a PhD student at the UK Medical Research Council’s National Institute of Medical Research and a resource team member for Health and Development Networks Stop-TB eForum.