By: Andy Extance
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
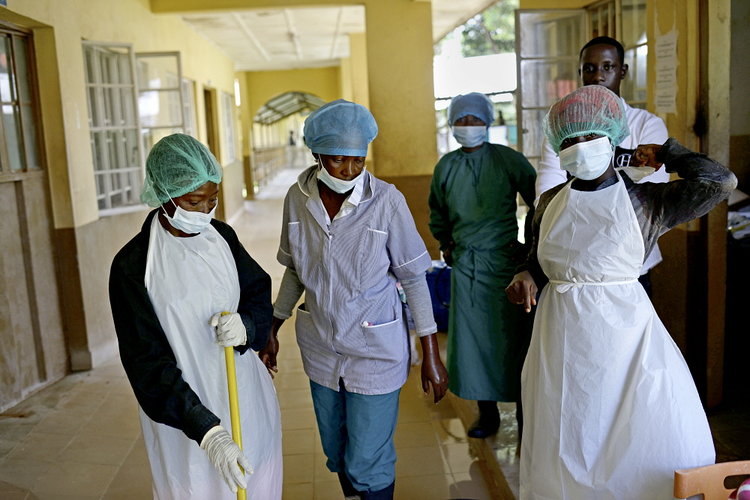
Efforts to save lives in the West African Ebola outbreak have been undermined by a failure to involve local people more closely in communication about treatment and ethical decisions about trials, says a report published last week (17 February).
The report’s authors, who are all involved in Ebola vaccine work, made recommendations focusing on Ebola vaccine research, manufacturing and the process of getting vaccine approval in the developed world. They were convened by UK medical research funder the Wellcome Trust and the Center for Infectious Disease Research and Policy at the University of Minnesota, United States.
Considering real human and social factors is vital for stemming the Ebola outbreak, says Clement Adebamowo, the chairman of the Nigerian National Health Research Ethics Committee and one of the report’s 26 advisers.
“Often, the many challenges and the desire for speed, efficiency and cost reduction in the attainment of scientific goals lead to the deprioritisation [of community involvement],” he says.
“Without appropriate and good quality community engagement, we may not succeed with our scientific interventions.”
The report found that Guinea, Liberia and Sierra Leone were unable to respond robustly to the epidemic due to their “struggling economies, civil wars, and the toll of Ebola”, which killed a lot of those at the frontline of the response. But the report says these problems were exacerbated by outside agencies underestimating the role communities play, for example in overcoming suspicions about treatments.
“Early communication about Ebola could have been done better in almost all affected countries,” Adebamowo says. “Failure to appropriately emphasise strong community engagement and communication strategies probably contributed to the ultimate scope of the epidemic.”
The report observed a lack of local involvement in organising clinical trials and related ethical decisions. It highlights that the Pan-African Bioethics Initiative, which might have helped organise such involvement, is struggling to obtain funding.
Adebamowo says this initiative should do more to attract “membership dues, support from industry, universities, research organisations, funding agencies and NGOs” to strengthen its presence in Ebola-affected countries.
“In Africa, research ethics is still in a nascent stage, but as research activities grow, we can expect that researchers, research sponsors and other stakeholders will appreciate the value of ethics,” he says.
Kevin Behrens, a bioethicist at the University of the Witwatersrand, South Africa, praises the report’s desire for local involvement. “However, this must mean that African stakeholders have a decisive role, not just that they will be consulted, but possibly overridden,” he warns.
> Link to Recommendations for accelerating the development of Ebola vaccines