By: Smriti Mallapaty
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
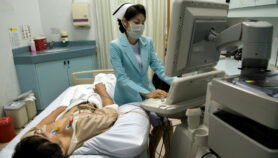
[SYDNEY] Strains of the visceral leishmaniasis parasite discovered in the central-eastern hills of Nepal have a different genetic and metabolic profile from the dominant population in the region, which could impact on the elimination of the disease, a study finds.
Scientists who conducted the study, published April in Infection, Genetics and Evolution, say the differences could affect the severity of infection with the Leishmania donovani parasite, which is transmitted by sandflies.
“We wanted to warn the community about the new strains as they could have implications for diagnosis and treatment”
Jean-Claude Dujardin, Institute of Tropical Medicine
“We wanted to warn the community about the new strains as they could have implications for diagnosis and treatment,” says Jean-Claude Dujardin, a biologist at the Institute of Tropical Medicine, Antwerp and lead author of the paper.
Visceral leishmaniasis, or kala-azar, is the second-most deadly parasitic disease after malaria, afflicting some 300,000 individuals a year and killing an estimated 30,000 of the poorest people in the world. The parasites attack the bone marrow, liver and spleen (hence the term visceral). Most cases occur in Bangladesh, India and Nepal, where a transnational effort led by the WHO aims to eliminate the neglected tropical disease by 2020.
In 2016, Dujardin was part of a team that sequenced more than 200 samples of L. donovani isolated from the South Asian region. They identified a dominant population, as well as two genetically distinct subpopulations, one found in Sri Lanka, and another, nicknamed yeti, in Nepal.
Dujardin and his colleagues wanted to find out just how unique the yeti strains were. The team compared the genomes and metabolite production of five yeti strains with four strains of the dominant group. The researchers observed significant differences in genes and metabolites related to virulence (harmfulness) of the disease. This, to the researchers, raised the possibility of other divergences more directly relevant to diagnosis and treatment.
“We do not have a good animal model for visceral leishmaniasis, so it’s always going to be unclear how the genetic and metabolic differences translate into pathogenicity, drug susceptibility and other key processes determining transmission and disease,” says Graham Medley, professor of infectious disease modelling at the London School of Hygiene and Tropical Medicine.
“On the whole, I have doubts about the success of the elimination programme,” comments Roy.
This piece was produced by SciDev.Net’s Asia & Pacific desk.