By: Claudia Caruana
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
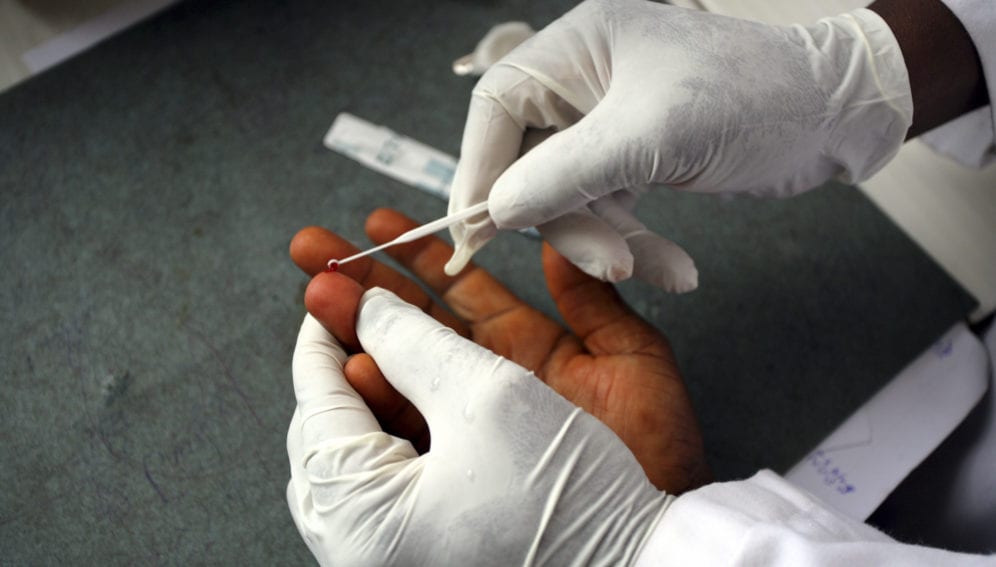
[NEW YORK] A team of researchers has adapted an existing test for falciparum malaria — the BinaxNOW malaria kit — to predict a dangerous complication that can arise after the Plasmodium falciparum parasite is cleared from patients' blood.
While most patients who receive the drug of choice (artemisinin artesunate) against malaria recuperate, a small number (less than 5 per cent) develop a serious and often fatal anaemia called post-artesunate delayed haemolysis (PADH) within a month of treatment.
“The diagnostic test measures the remnants of the malaria protein HRP2 on red blood cells.”
Pierre Buffet, Paris Descartes University
Clinicians have been unable to predict who might develop PADH which can lead to kidney failure and death, says Papa Alioune Ndour, a member of the research team from the National Institute of Blood Transfusion under the Paris Descartes University.
Working on anecdotal evidence, the researchers repurposed the BinaxNow malaria kit and found significant degree of accuracy in detecting PADH. The team’s findings were published 5 July in Science Translational Medicine.
Pierre Buffet, head of the RBC (red blood cell) research team at the faculty of medicine, Paris Descartes University, says the plan is to use the same test three days after the start of treatment, instead of the current recommendations for 28-day monitoring of all malaria patients.
“The diagnostic test measures the remnants of the malaria protein HRP2 (Histidine-Rich Protein 2 which is secreted by the parasite into the host’s blood stream) on red blood cells,” says Buffet.
According to Buffet, the modified diagnostic test predicted PADH with 89 per cent sensitivity and 73 per cent specificity. Blood samples for the test were obtained from 95 infected Bangladeshis in Asia and 53 French travellers who were infected in Africa but returned home and received treatment.
Buffet says the direct cost of prediction would now be acceptable (even in low-income countries) compared to conventional enumeration of ‘pitted’ RBCs by flow cytometry, a long-known cell biology process which requires heavy and expensive equipment.“Our first step is to perform a multisite study to confirm predictive thresholds and impact, predominantly in African children,” Buffet says.
The US Centers for Disease Control and Prevention (CDC) currently recommends that patients treated for high-density malaria infections be evaluated 7—10 days later to check for this potential problem and be offered treatment when necessary.
S. Patrick Kachur, malaria branch chief at the Atlanta-based CDC, notes that any test that can reliably determine the parasite burden should be capable of identifying this increased risk associated with high levels of malaria infection.
This piece was produced by SciDev.Net’s Asia & Pacific desk.