By: Claudia Caruana
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
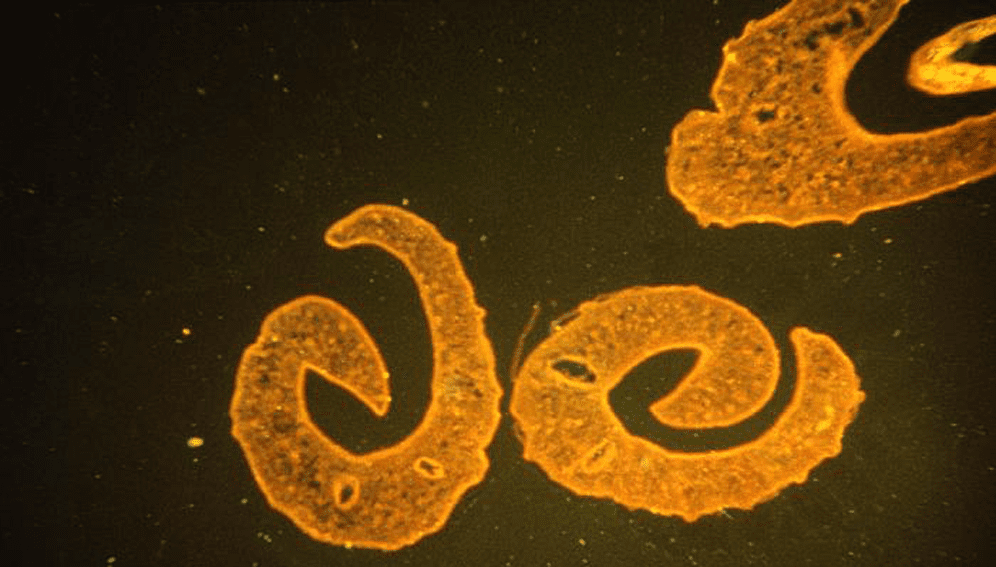
David Diemert, an associate professor of medicine at George Washington School of Medicine and Health Sciences, has long been working on vaccines against helminthic parasites such as hookworm and liver fluke. While the diseases caused by these parasites affect an estimated one billion people worldwide (see table), little progress has been made to remedy the situation.
“Developing vaccines for worms is additionally challenging. These are large organisms compared to viruses or bacteria, which adds complexity to the vaccine development process”
David Diemert, George Washington School of Medicine and Health Sciences
That is about to change, says Diemert, lead author of a paper on the difficulties of developing vaccines against parasitic worms, published this month (September) in Trends in Parasitology. He reveals that he now has hookworm and schistosomiasis vaccine candidates under clinical testing.
“Creating a helminth vaccine is arduous. There are challenges at all stages. But at many points, we were responsible for creating novel testing, formulation and manufacturing processes in order to advance to the next step in our clinical trials,” Diemert tells SciDev.Net in this interview.
Table. Major human helminthiases and their global prevalence and distribution
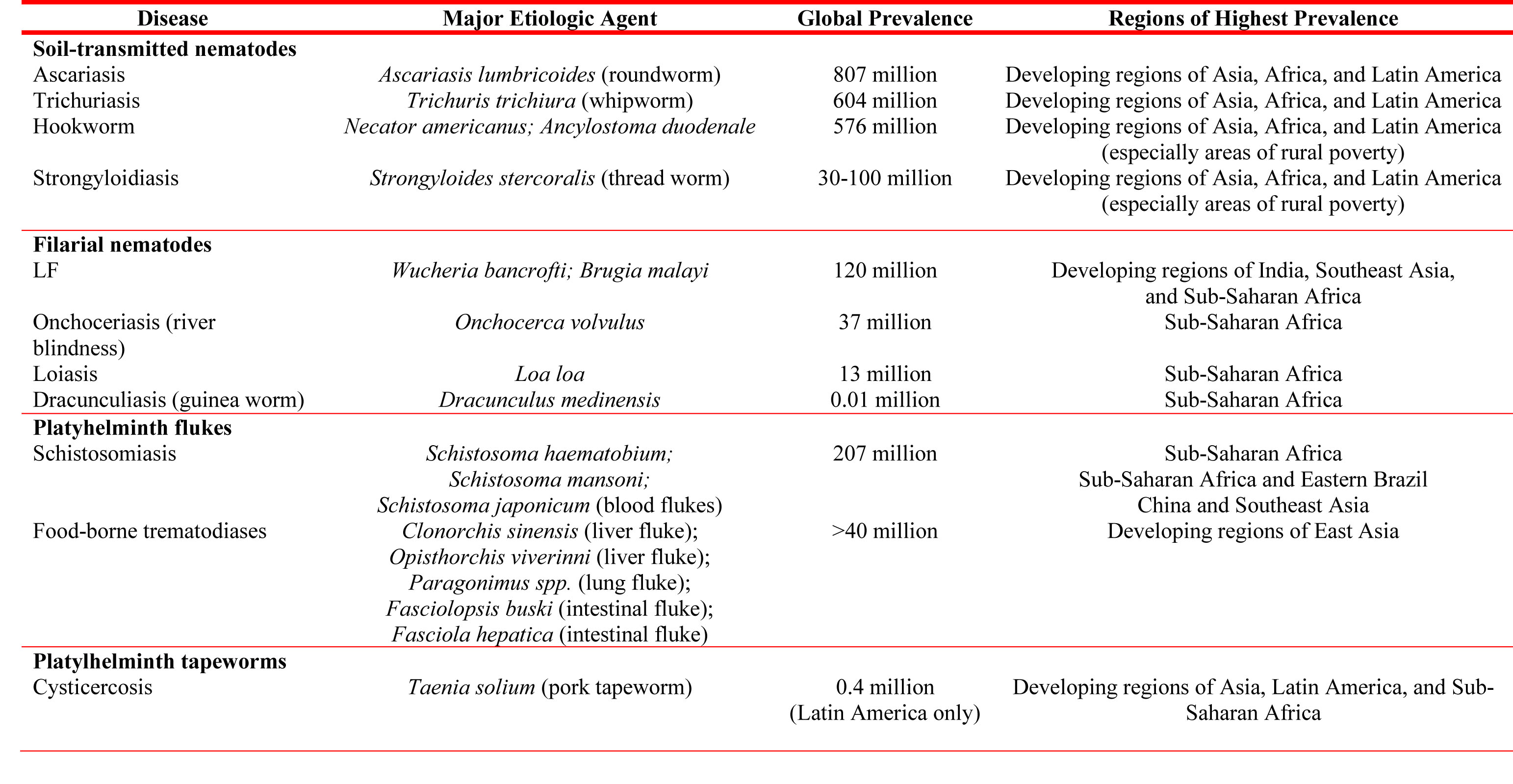
Source: The Journal of Clinical Investigation
How close are we to having vaccines to prevent these parasitic diseases?
Vaccines for hookworm are close to licensing but it will still be several years before an application could be made to regulatory agencies. We’ve just started a phase two proof-of-efficacy trial of hookworm vaccines here at George Washington. If successful, a pivotal phase three trial will be needed in the field, and these take a minimum of 2—3 years to complete.
Where are these vaccines being tested now? Are you primarily working on vaccines for children or adults as well?
Vaccines for hookworm are being currently tested at George Washington and in Gabon, Africa. A vaccine for schistosomiasis is currently underway in Brazil. Vaccines for both diseases are being developed mainly for children since the impact of the infections is seen mostly in this age group.
Can you describe your efforts at George Washington with your Texas partners? Do their researchers have a different focus?
Our efforts at George Washington are closely linked with those at Baylor College of Medicine in Texas. Together, we form a "product development partnership”. At George Washington, we manage the clinical development and immunology testing for the programme, whereas at Baylor the focus is more on the vaccine manufacturing process development, preclinical quality testing of the vaccines, and the regulatory support for the programme.
There seems to be less interest in these diseases than other diseases in the developing world such as malaria.
These diseases affect only those living in poor, rural areas of the tropics. Also, they cause mostly chronic health effects and not acute mortality, so they have a more insidious and hard-to-measure impact on health. There have, however, been recent reports of possible ongoing transmission in poor, rural areas of the south-eastern US in communities without adequate sanitation.
Why has there been so much difficulty in developing these vaccines?
A lot of the difficulty stems from a lack of funding. Developing vaccines for worms is additionally challenging, however. These are large organisms compared to viruses or bacteria, which adds complexity to the vaccine development process.
What lessons have you learned from developing these difficult vaccines?
In our most recent paper describing our work, we have learnt five lessons:
• Allow for ample time and resources during process development and manufacturing of helminth vaccines, with a focus on ability to scale-up to industrial-level manufacturing.
• Develop vaccine potency assays that do not rely on the traditional immunisation-challenge potency model (as used for vaccines against pertussis, diphtheria, and rabies) as helminth pathogens neither induce reliable mortality in an animal model nor do these vaccines induce sterilising protection.
• Overcome the ‘deficient acquired immunity’ that occurs during chronic helminth infection in order to attain sufficient vaccine immunogenicity in these individuals.
• Accelerate vaccine efficacy testing in humans through use of controlled human infection models as currently established at George Washington for controlled human hookworm infection.
There actually is a vaccine for Schistosoma japonicum that has been developed for cattle since they are a reservoir of infection. If transmission to cattle could be blocked, it could potentially reduce subsequent transmission to humans.
This piece was produced by SciDev.Net’s Asia & Pacific desk.