By: Sam Otieno
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
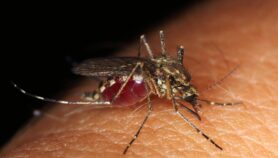
The rapid and widespread resistance to the malaria drug, sulfadoxine-pyrimethamine (SP) in Tanzania could undermine improvement in malaria treatment during pregnancy, a study says.
The study conducted in Tanzania established that the prevalence of SP-resistant malaria parasites remains high, putting pregnant mothers and the neonates at considerable health risk.
According to the researchers from Belgium and Tanzania, their aim was to determine the status of resistance to the drug six years after it was abandoned as a first line regimen for treating uncomplicated malaria in the country.
“Authorities should come up with appropriate interventions in the endemics areas so as to reduce the parasite load.”
Abdoulaye Djimdé, University of Science, Techniques and Technologies of Bamako, Mali
The drug is rather used for intermittent preventive treatment of malaria, during which pregnant women receive the medicine at routine prenatal visits regardless of whether they are infected with malaria.
Through cross-sectional sampling, they selected 264 patients who were at least six months old, lived in one of three regional sites — Muheza, Muleba and Nachingwea — in mainland Tanzania and went to health facilities for treatment of malaria from May to August 2013.
The researchers analysed the blood samples of the patients for high amounts of the double mutant genes Pfdhfr – Pfdhps, which indicate drug resistance.
“The prevalence of Pfdhps, K540 E at Muheza was significantly higher (95.4 per cent) compared to the other sites,” the authors note in Malaria Journal published last month (5 November).
The researchers add that the WHO recommends SP should not be used for intermittent prevention of malaria in pregnancy if the prevalence of this gene is more than 95 per cent.
Vito Baraka, a co-author of the study and a research scientist at the Tanzania-based National Institute for Medical Research (NIMR), tells SciDev.Net: “Due to the changing [nature] of malaria epidemiology in Tanzania, we wanted to know the dynamics of the transmission of the sulfadoxine-pyrimethamine resistance in the country.”
According to Baraka, intermittent preventive treatment of malaria is beneficial in reducing episodes of pregnancy-associated malaria, risk of anaemia, low birth weight, neonatal mortality and other associated consequences.
But Baraka says the findings suggest an urgent need to conduct efficacy and safety studies to determine alternative drugs for intermittent preventive treatment of malaria in pregnancy.
“In addition, we need to increase control efforts [such as] early screening by rapid diagnostic tests, bednets coverage and vector control in these settings where the resistance is at alarming levels,” Baraka tells SciDev.Net.
Barak adds: “Currently there are ongoing clinical trials to evaluate alternatives for intermittent preventive treatment of malaria in pregnancy using sulfadoxine-pyrimethamine in Ghana, Kenya, Mali and Zambia that offer an optimism that we can replace SP with a more effective drugs such as co-trimoxazole, dihydroartemisinin-piperaquine and artemether-lumefantrine, if proved.”
Abdoulaye Djimdé, head of the Molecular Epidemiology and Drug Resistance Unit of the University of Science, Techniques and Technologies of Bamako, Mali, agrees that pregnant women are not benefiting from SP, and there is need to change the treatment now to artemisinin-based combination therapies (ACTs) used as first-line regimen for malaria treatment.“However, authorities should come up with appropriate interventions in the endemics areas so as to reduce the parasite load,” Djimdé says, citing a need for public education on how to prevent mosquito bites through destroying the breeding sites, and encouraging pregnant women to visit clinics as required.
This piece was produced by SciDev.Net’s Sub-Saharan Africa English desk.
References
Vito Baraka and others High-level Plasmodium falciparum sulfadoxine-pyrimethamine resistance with the concomitant occurrence of septuple haplotype in Tanzania (Malaria Journal, 5 November 2015)