By: Crispin Maslog
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
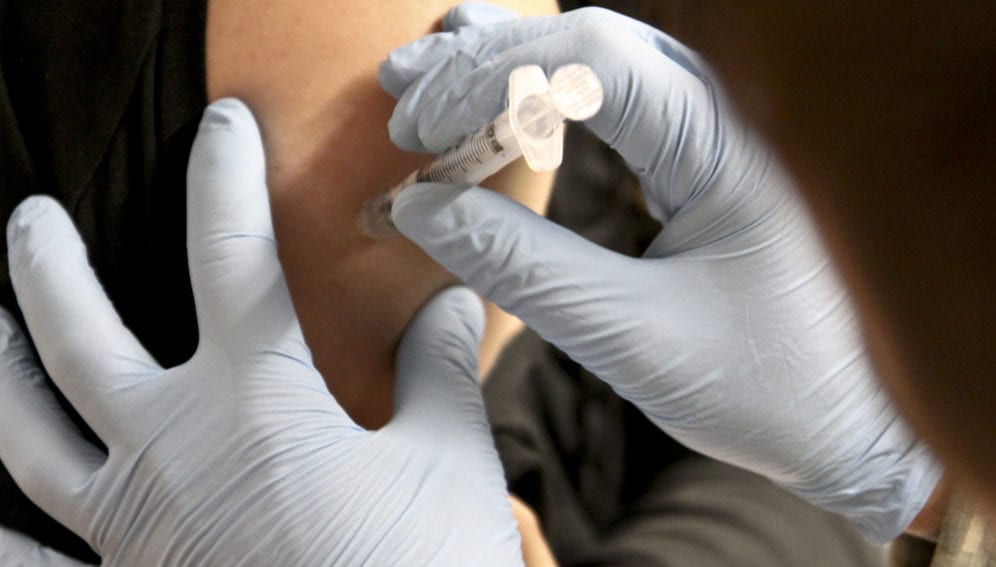
Asia’s first dengue vaccination programme has been launched and is picking up steam in the Philippines despite misgivings in some local health quarters.
The Philippines became the first Asian country to license Sanofi Pasteur’s dengue vaccine, Dengvaxia, last 22 December, following Mexico’s approval of the vaccine on December 11.
Dengvaxia is a tetravalent vaccine taken in three shots at six-month intervals and is recommended for individuals aged 9 to 45. The challenge with dengue is to develop a vaccine that is safe and effective against all four dengue strains. Dengvaxia is assessed to be 90 per cent effective.
Three Philippine government ministries — the Department of Health (DOH), Department of Education, and Department of Interior and Local Government — collaborated in a major launch of the vaccine in Metro Manila on 4 April. [1]
President Benigno Aquino III was also present when DOH secretary Janette Garin administered the injection on one of the first children to be vaccinated.
Questions about prequalification
The Philippines went ahead with the school-based mass vaccination programme even before it was prequalified by the WHO. Prequalification is a standard procedure by WHO to ensure that vaccines purchased by the UN and government procurement agencies are safe and effective.
This has led some medical doctors to raise doubts about the early launch of the mass vaccination programme. The doubting doctors include Teodoro Herbosa, former DOH undersecretary, and Anthony Leachon, board member of the Philippine Health Insurance (PhilHealth). They oppose the implementation of the DOH’s 3-billion peso (US$64 million) mass immunisation programme without WHO prequalification.
However, the WHO Strategic Advisory Group of Experts on immunisation has reviewed Dengvaxia (CYD-TDV) in April 2016 and recommended introduction of the vaccine in countries or places where dengue is endemic even while awaiting WHO prequalification.
One post-vaccination death reported
As of 24 April, the DOH has vaccinated 204,397 students in grade schools in the Manila metropolis, out of the 279,393 students whose parents have expressed consent to the vaccination. [3]
The DOH reported 362 cases of Adverse Event Following Immunisation (AEFI). This is medical jargon for side effects to the vaccine, which include fever, headache, dizziness, vomiting and rashes.
The percentage of students that developed side effects, however, is minuscule — less than one percent of the total number immunised.
The death of an 11-year-old boy was reported after his immunisation on 31 March although he was screened and assessed by a health doctor before vaccination. He developed fever and diarrhoea on 3 April. He was in and out of different hospitals following that, experiencing difficulty of breathing, fever and cough. He was diagnosed with pneumonia, severe congenital heart disease and electrolyte imbalance. On 11 April the patient had cardiac arrest.
The case was presented to the DOH National Adverse Event Following Immunization Committee for review, after which the committee said it was coincidental that the boy had cardiac arrest after immunisation.
WHO support
The WHO has endorsed the decision of the Philippines to go ahead with its dengue mass vaccination programme based on its own scientific appraisal of the clinical research that preceded adoption of the vaccine.
The WHO country representative in the Philippines, Gundo Weiler, said in a press conference, that they support the Philippine dengue vaccination programme. [4]
Weiler explained that WHO’s Strategic Group of Experts on Immunization meets twice a year to vet new medical products like drugs and vaccines and advise WHO on what products to approve. The WHO formal prequalification guidelines and recommendations on the new dengue vaccine are expected to be released in May.
Dengue vaccine pioneers
The Philippines is the first country to implement dengue vaccination in schools and aims to immunise one million grade school children aged nine years and above in the next few years.
Dengue is highly endemic in the Philippines. It ranked first in highest dengue incidence in the Western Pacific region from 2013 to 2015.
There are some 400 million dengue virus infections globally each year. The Asia-Pacific region has the highest incidence of dengue in the world, accounting for more than 70 per cent of dengue cases. South-East Asia alone had an average of 2.9 million dengue cases and 5,906 deaths from 2000 to 2010. [5]
The Philippines has taken a bold step to try this new dengue vaccine in Asia on such a massive scale. It joins only two other countries so far, Mexico and Brazil in South America, in daring to be among the pioneers.
In the face of data that dramatise the dengue danger, Philippine health officials have decided it is worth taking the risk. The reward looks to be great — a defense against a disease that is not to be taken lightly. The next steps should be against the Zika and Ebola viruses.
Crispin Maslog is a Manila-based consultant for the Asian Institute of Journalism and Communication. A former journalist, professor and environmental activist, he worked for the Press Foundation of Asia and the International Rice Research Institute.
This piece was produced by SciDev.Net’s South-East Asia & Pacific desk.
References
1. Department of Health Press Release PH successfully launched the first dengue vaccines in public schools (DOH, 13 April 2016)
2. Department of Health Press Release DOH releases latest dengue immunisation coverage (DOH, 25 April 2016)
3. Department of Health National Capital Region Office WHO experts group clears administration of dengue vaccines in areas with high dengue rates (DOH, 18 April 2016)
4. Sabin Vaccine Institute Statement by the Dengue Vaccine Initiative on the Philippines Regulatory Approval of Sanofi Pasteur’s Dengue Vaccine, Dengvaxia (Sabin, 23 December 2015)