By: Crispin Maslog
Send to a friend
The details you provide on this page will not be used to send unsolicited email, and will not be sold to a 3rd party. See privacy policy.
A forgotten tool to fight antibiotic resistance may avert the threat of a post-antibiotic era — the possibility of antibiotics becoming ineffective against bacteria and people dying of serious or even common infections.
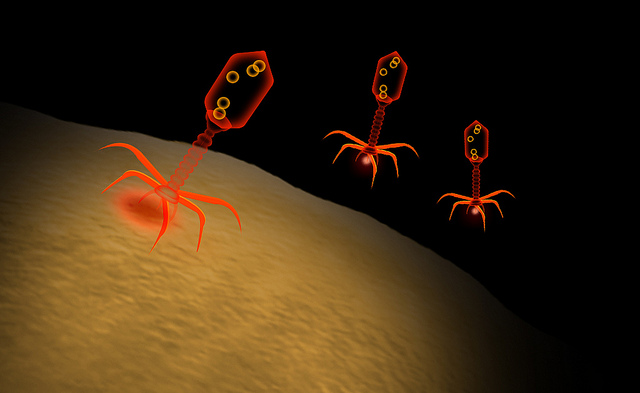
This forgotten tool is bacteriophage therapy, which was first discovered by British bacteriologist Frederick Twort in 1915. In the 1930s, phage was used in many countries to treat millions of patients against infectious diseases like cholera, bacillary dysentery and furuncles.
But once penicillin was discovered in the 1940s, phage therapy fell by the wayside in the West but their use has continued in Eastern Europe and in the former Russian republics.
A bacteriophage, or phage for short, is a virus that infects and replicates inside a bacterium. The term bacteriophage comes from bacteria and the Greek phagein (to devour). A virus is a small infectious agent that replicates only inside the living cells of other organisms. In phage therapy, a virus literally eats the bacteria.
David Trudil, advisor to the George Eliava Institute of Bacteriophages in Tbilisi, Georgia, is spearheading the revival of interest in the century-old biophage therapy in response to the growing threat of drug-resistant pathogens.
In April, the WHO, in a 257-page report of 114 countries, sounded the alarm for a post-antibiotic era.
“Drug-resistant pathogens are a growing menace to all individuals regardless of age or socio-economic background. They endanger industrial societies as well as less developed nations and are causing problems in hospitals, homes and more,” explained Trudil, who is also president of NHDetect Corp., a biotech firm. [1]
In the recent annual convention of the Philippine Society for Microbiology in Manila (16-17 July), Trudil told the meeting it is time to bring bacteriophage therapy back, arguing that it is now the most promising approach to treat bacterial diseases that have become resistant to antibiotics.
Trudil’s recent phage-based projects include treatment, therapies and disease prevention in humans, and agricultural and environmental applications.
Origins of phage therapy
The use of phages as a therapy for infectious bacterial diseases was first seriously studied by Felix d'Hérelle, a bacteriologist at the Pasteur Institute in Paris, during the First World War. [2]
D'Herelle isolated phages from the faeces of French soldiers sick with bacillary dysentery. He observed that the microbe appeared in the stools of patients just before they began to recover from the disease. He concluded that these phages had something to do with the healing process of bacillary dysentery.
A Georgian, George Eliava, was making similar discoveries. He travelled to the Pasteur Institute in Paris where he met d'Hérelle, and in 1923 he founded the Eliava Institute in Tbilisi, Georgia, devoted to the development of phage therapy.
In the United States during the 1940s, commercialisation of phage therapy was undertaken by the large pharmaceutical company, Eli Lilly. But early research studies on phages were often unreliable.
Phage therapy survived mostly in Eastern Europe side by side with antibiotics. Only after the fall of the Soviet Union did Western scientists remember the parallel medicine from behind the Iron Curtain — their interest stimulated by the growing problem of antibiotic resistance.
During the Cold War era, political barriers prevented scientific knowledge from being translated and shared across the world. It was only in 2009 that a summary of these publications was published in English.
There is an extensive library and research centre at the Eliava Institute and phage therapy is today a widespread form of treatment in that region.
Phage therapy vs antibiotics
Phage therapy has several advantages over antibiotics. One of them is that phages are very specific and do not harm the useful bacteria that live in and on the body. Antibiotics, on the other hand, also attack harmless bacteria in the human body systems.
While bacteria also develop resistance to phages, it is very much easier to develop new phages than new antibiotics. A few weeks compared to years are needed to develop new phages for new strains of resistant bacteria. As bacteria evolve resistance, the relevant phages also evolve alongside. When super bacterium appears, the super phage is ready to attack it.
Phages also do not develop secondary resistance, which happens often in antibiotics. With the increasing incidence of antibiotic-resistant bacteria and slow development of new antibiotics, phage treatment therefore comes in handy.
In one of the few clinical studies on phage therapy, the Phage Therapy Center in Tbilisi, Georgia, used staphylococcus aureus phages to treat patients for purulent disease of the lungs and pleura. One group received phages while the other group received antibiotics.
Complete recovery was observed in 82 per cent of the patients in the phage-treated group compared to 64 per cent of the patients in the antibiotic-treated cluster.
Because phages are not antibiotics, they do not have as many uses that contribute to bacterial evolution of resistance, which range from treating animal and plant diseases to increasing food-animal growth rates and improper use in treating human diseases. Phages do not contribute to antibiotic resistance and therefore could help extend the usefulness of conventional antibiotics.
Another advantage is that phages are natural products and will not draw the public criticism of genetically modified organisms.
In an era where antibiotic-resistant bacterial infection is on the rise, this forgotten tool called phage therapy may just be the answer people are looking for.
Crispin Maslog is a former journalist and science journalism professor at the University of the Philippines Los Baños and director of the Silliman School of Journalism, Philippines. He is a consultant of the Asian Institute of Journalism and Communication and board chairperson of the Asian Media Information and Communication Centre, both based in Manila.
This article has been produced by SciDev.Net's South-East Asia & Pacific desk.
References
[1] David Trudil Bacteriophage, an Emerging Tool to Combat Antibiotic Resistance, Plenary Lecture, 44th PSM Annual Convention and Scientific Meeting, 16-17 July, 2015, Metro Manila
[2] Wikipedia Phage therapy (Accessed 8 August 2015)